Periodic Table Families Periodictable Ca

Periodic Table Families Periodictable Ca The periodic table of elements is a way of organising the vast array of different elements found in chemistry. the table is made up of rows, known as periods, and columns, known as groups or families. Periods, groups, and families of the periodic table defined with a labeled diagram. how many are there. what are their names. learn their properties with examples.
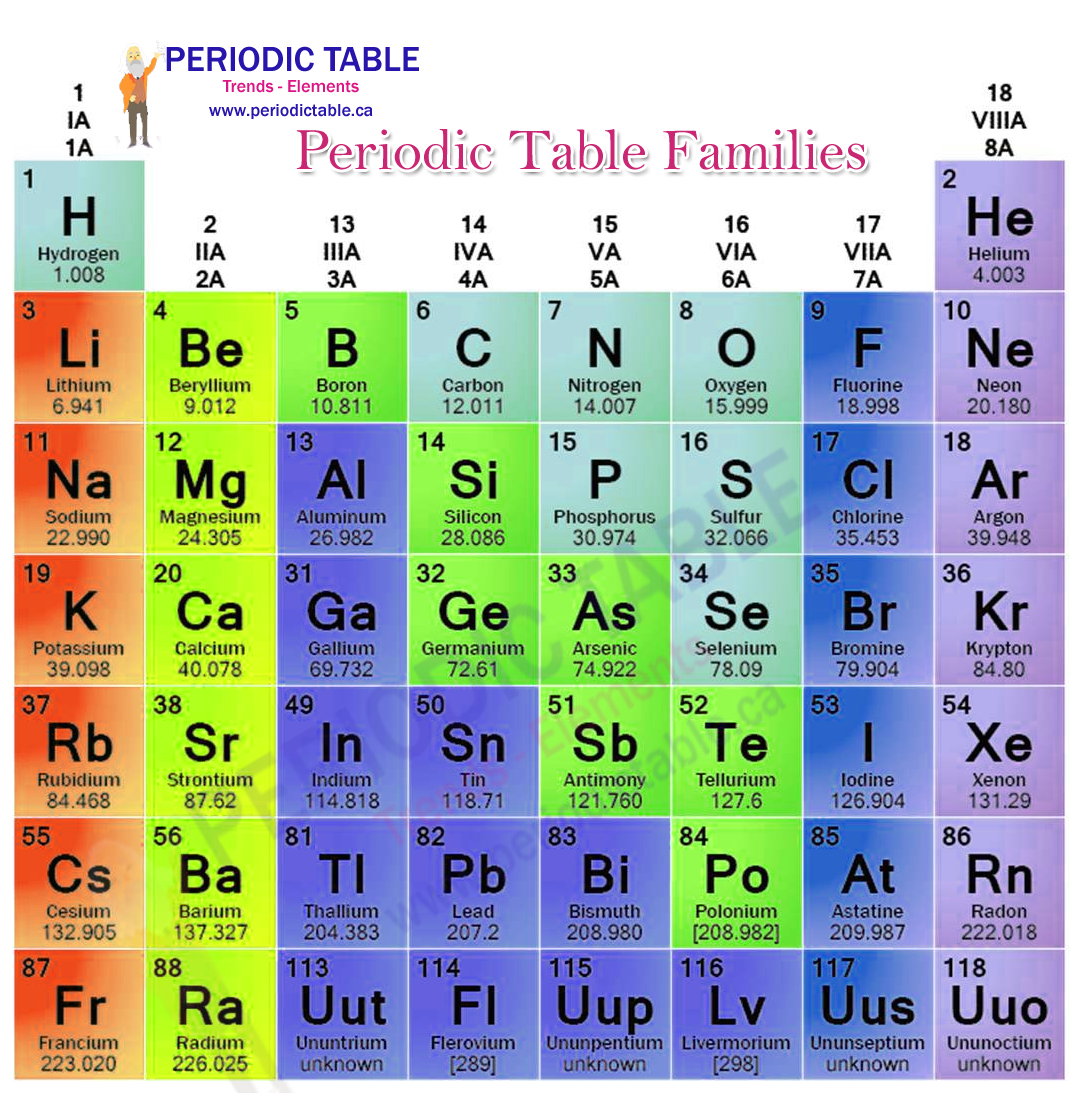
Periodic Table Families Periodictable Ca Elements may be categorized according to element families. this is an explanation of the different element families, with descriptions of each one. Learn about element families on the periodic table. learn how to identify each family and see its elements and properties. Explore how the periodic table organizes elements by increasing atomic number and reveals repeating patterns in properties grouping metals, nonmetals, and metalloids into families like alkali metals, halogens, and noble gases, thus allowing for for predictive insight into chemical behavior. Calcium (ca) is in group 2, for it contains two valence electrons. in the old iupac system the letters a and b were designated to the left (a) and right (b) part of the table, while in the cas system the letters a and b are designated to main group elements (a) and transition elements (b).
Periodic Table Families Labelled Diagram Explore how the periodic table organizes elements by increasing atomic number and reveals repeating patterns in properties grouping metals, nonmetals, and metalloids into families like alkali metals, halogens, and noble gases, thus allowing for for predictive insight into chemical behavior. Calcium (ca) is in group 2, for it contains two valence electrons. in the old iupac system the letters a and b were designated to the left (a) and right (b) part of the table, while in the cas system the letters a and b are designated to main group elements (a) and transition elements (b). Interactive periodic table with element scarcity (sri), discovery dates, melting and boiling points, group, block and period information. The vertical columns on the periodic table are called groups or families because of their similar chemical behavior. all the members of a family of elements have similar chemical properties. The vertical columns on the periodic table are called groups or families because the elements in the column exhibit similar chemical behaviors. the groups on the periodic table also have similar electron configurations. Calcium is a chemical element of the periodic table with chemical symbol ca and atomic number 20 with an atomic weight of 40.0784 u and is classed as alkaline earth metal and is part of group 3 (scandium group).
Comments are closed.