Performance Qualification Pdf

Performance Qualification Protocol Pdf Sterilization Microbiology This document provides a template for generating a performance qualification (pq) protocol. To establish the methodology for the performance qualification of purified water system, which is used for generation, storage and distribution of purified water in solid oral and liquid oral block including quality control department.

Performance Qualification Pq In Pharmaceutical Industry The purpose of this process performance qualification protocol is to produce consistent quality of product by given sets of equipments & process conditions by using batch manufacturing record (bmr). All personnel involved in this qualification must complete the table below to provide identification of all the signatures initials used in this document. Table 1 shows the most common approaches to satisfying pq testing requirements for hplc instruments. similar approaches to the three options in the table have been used for all chromatography instruments. each of these choices has apparent advantages and disadvantages. Pq performance qualification sds software design specification sop standard operating procedure spreadsheet generic term for application containing rows and columns of cells, with functions to manipulate data within those cells.
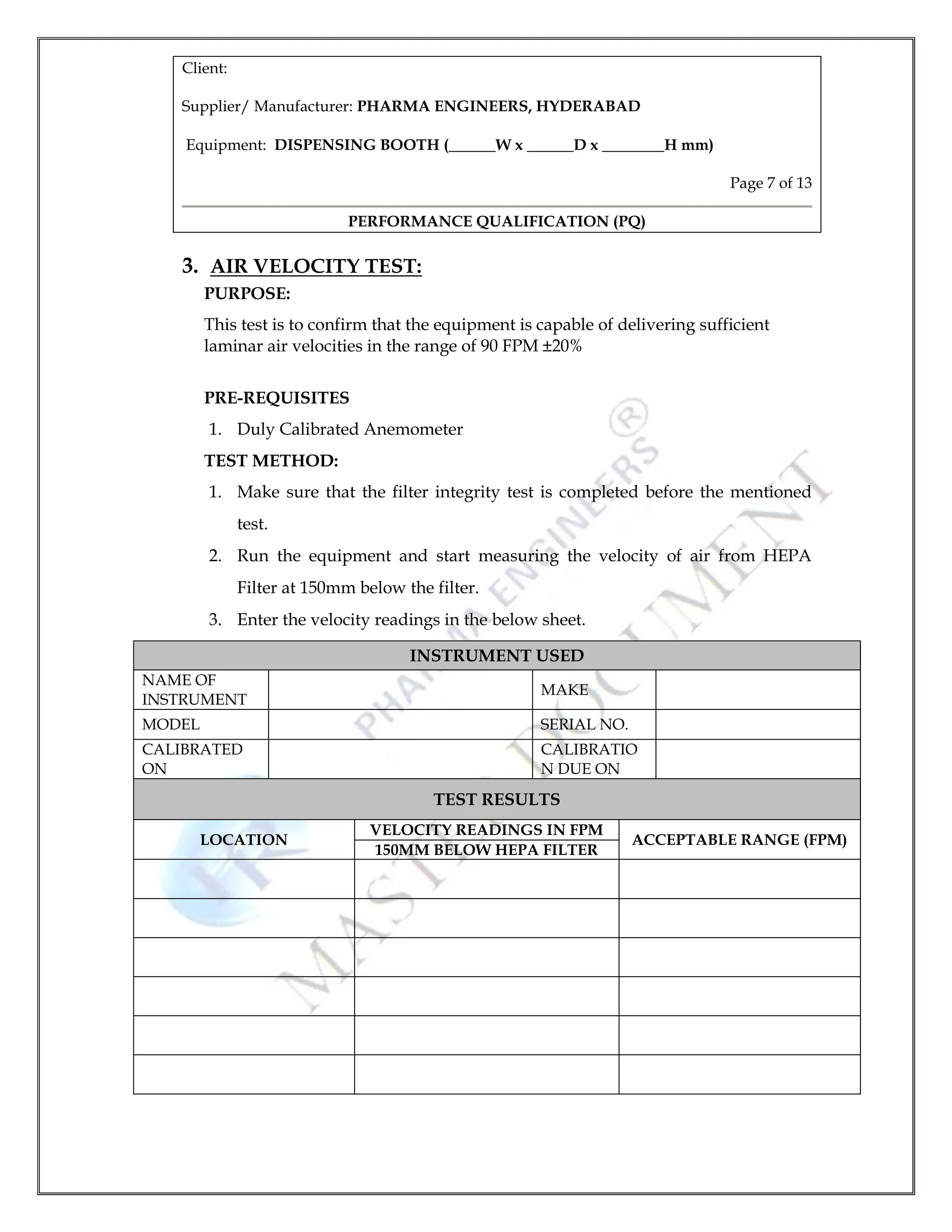
8 4 Performance Qualification Protocol For Dispensing Booth 1 Pdf Table 1 shows the most common approaches to satisfying pq testing requirements for hplc instruments. similar approaches to the three options in the table have been used for all chromatography instruments. each of these choices has apparent advantages and disadvantages. Pq performance qualification sds software design specification sop standard operating procedure spreadsheet generic term for application containing rows and columns of cells, with functions to manipulate data within those cells. Pdf | on jun 1, 2022, faiz nasrullah published protokol kualifikasi kinerja (performance qualification) steam air cycle (autoclave) | find, read and cite all the research you need on. All employees involved in this pq must be trained to their work. training records must be kept on file. Abbreviations and definitions pq: performance qualification; documented verification that the integrated system or subsystem functions as intended, in its normal operating environment. Conditional approval to proceed to the next qualification stage can be given where certain acceptance criteria or deviations have not been fully addressed and there is a documented assessment that there is no significant impact on the next activity.
Comments are closed.