Pdf Adaptive Trial Designs

Adaptive Trial Designs Litfl Ccc Research Pdf | on jul 7, 2016, deepak l. bhatt and others published adaptive designs for clinical trials | find, read and cite all the research you need on researchgate. For the purposes of this guidance, an adaptive design is defined as a clinical trial design that allows for prospectively planned modifications to one or more aspects of the design based on.

Pdf Adaptive Trial Designs Data from the published literature on study characteristics, types of adaptations, statistical analysis, stopping boundaries, logistical challenges, operational considerations and ethical considerations were extracted and summarized herein. We outline and discuss common characteristics and study modifications of adaptive trials and provide a practical planning guide for designing and interpreting adaptive clinical trials. Derstanding of adaptive trial designs and master protocols. this book is intended for those with minimal background in clinical trial research and to help readers grasp strengths and limitations of these novel designs and appl. Download or read book adaptive design of clinical trials with interim selection of treatment arms written by yueh wen liao and published by . this book was released on 2012 with total page ? pages. available in pdf, epub and kindle. book summary: when developing new drugs, phase i and ii trials are commonly conducted to determine the dose of the new treatment in preparation for the subsequent.

Adaptive Trial Designs World Bi Derstanding of adaptive trial designs and master protocols. this book is intended for those with minimal background in clinical trial research and to help readers grasp strengths and limitations of these novel designs and appl. Download or read book adaptive design of clinical trials with interim selection of treatment arms written by yueh wen liao and published by . this book was released on 2012 with total page ? pages. available in pdf, epub and kindle. book summary: when developing new drugs, phase i and ii trials are commonly conducted to determine the dose of the new treatment in preparation for the subsequent. Applied clinical trials is the thought leader in pharmaceutical drug development operations and clinical trial design, management, and monitoring. We explain the basic rationale behind adaptive designs, clarify ambiguous terminology and summarise the utility and pitfalls of adaptive designs. we discuss practical aspects around funding, ethical approval, treatment supply and communication with stakeholders and trial participants. To illustrate implications for sample size, study dura tion, and power that could be obtained using adaptive trial strategies, we compare the operating character istics of the motivate and remind trials using the original design or outcome adaptive bayesian design. Adaptive designs for clinical trials of drugs and biologics guidance for industry0f.

Adaptive Trial Designs World Bi Applied clinical trials is the thought leader in pharmaceutical drug development operations and clinical trial design, management, and monitoring. We explain the basic rationale behind adaptive designs, clarify ambiguous terminology and summarise the utility and pitfalls of adaptive designs. we discuss practical aspects around funding, ethical approval, treatment supply and communication with stakeholders and trial participants. To illustrate implications for sample size, study dura tion, and power that could be obtained using adaptive trial strategies, we compare the operating character istics of the motivate and remind trials using the original design or outcome adaptive bayesian design. Adaptive designs for clinical trials of drugs and biologics guidance for industry0f.

Adaptive Design Clinical Trials To illustrate implications for sample size, study dura tion, and power that could be obtained using adaptive trial strategies, we compare the operating character istics of the motivate and remind trials using the original design or outcome adaptive bayesian design. Adaptive designs for clinical trials of drugs and biologics guidance for industry0f.
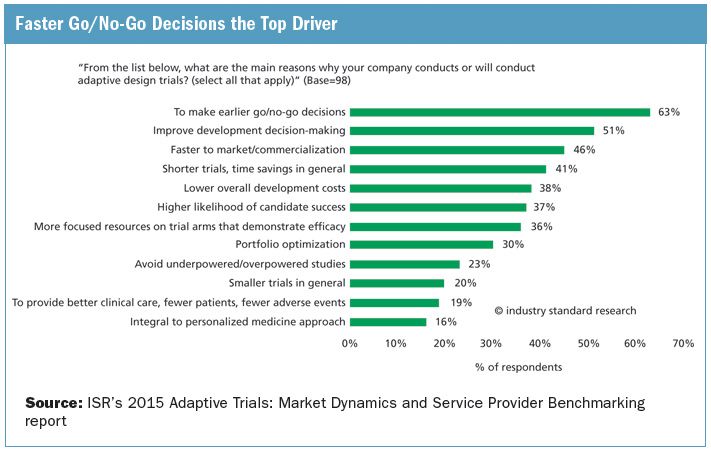
Adaptive Trial Designs Gaining Momentum
Comments are closed.