Pdf A High Throughput Sars Cov 2 Pseudovirus Multiplex Neutralization

Pdf A High Throughput Sars Cov 2 Pseudovirus Multiplex Neutralization Evaluating the neutralizing antibody titer following sars cov 2 vaccination is essential in defining correlates of protection. we describe an assay that uses single cycle vesicular stomatitis virus (vsv) pseudoviruses linking a fluorophore with a spike (s) from a variant of concern (voc). Evaluating the neutralizing antibody titer following sars cov 2 vaccination is essential in defining correlates of protection. we describe an assay that uses single cycle vesicular stomatitis virus (vsv) pseudoviruses linking a fluorophore with a spike (s) from a variant of concern (voc). using two fluorophores linked to two voc s, respectively, allows us to determine the neutralization titer.

Pseudovirus And Authentic Virus Neutralization A Sars Cov And Using two fluorophores linked to two voc s, respectively, allows us to determine the neutralization titer against two vocs in a single run. this is a generalizable approach that saves time. Evaluating the neutralizing antibody titer following sars cov 2 vaccination is essential in defining correlates of protection. we describe an assay that uses single cycle vesicular stomatitis virus (vsv) pseudoviruses linking a fluorophore with a spike (s) from a variant of concern (voc). Evaluating the neutralizing antibody titer following sars cov 2 vaccination is essential in defining correlates of protection. we describe an assay that uses single cycle vesicular stomatitis virus (vsv) pseudoviruses linking a fluorophore with a spike (s) from a variant of concern (voc). This article describes the development of a pseudovirus based neutralization assay for evaluating the neutralizing antibodies against emerging sars cov 2 variants in cost effective bsl 2 research conditions.

Antibody Neutralization Response Against Pseudoviruses Expressing Sars Evaluating the neutralizing antibody titer following sars cov 2 vaccination is essential in defining correlates of protection. we describe an assay that uses single cycle vesicular stomatitis virus (vsv) pseudoviruses linking a fluorophore with a spike (s) from a variant of concern (voc). This article describes the development of a pseudovirus based neutralization assay for evaluating the neutralizing antibodies against emerging sars cov 2 variants in cost effective bsl 2 research conditions. This article describes the validation of an in vitro pseudovirus based neutralization (pnt) assay to detect and quantitate neutralizing antibodies against sars cov 2 ancestral and variant strains (omicron ba.5 and omicron xbb.1.5) at novavax clinical immunology laboratories.
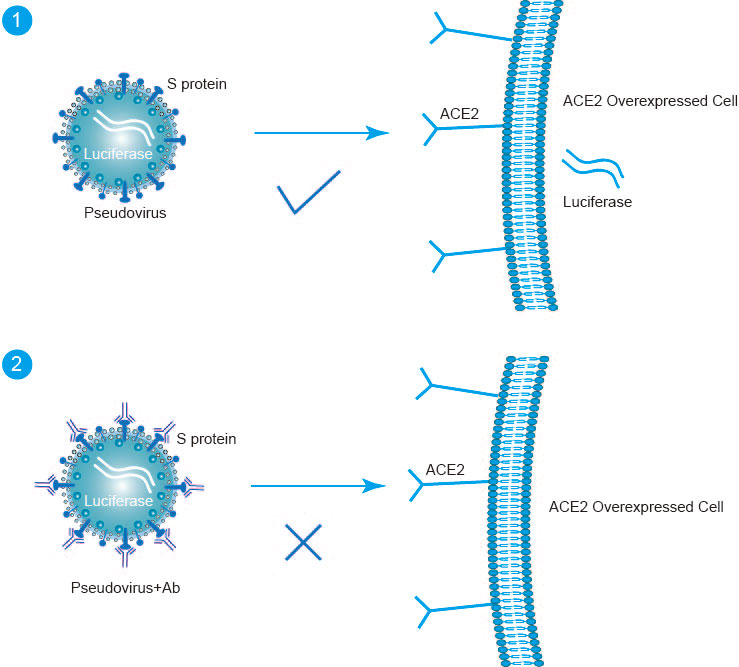
Sars Cov 2 Pseudovirus Neutralization Assay Creative Diagnostics This article describes the validation of an in vitro pseudovirus based neutralization (pnt) assay to detect and quantitate neutralizing antibodies against sars cov 2 ancestral and variant strains (omicron ba.5 and omicron xbb.1.5) at novavax clinical immunology laboratories.
Comments are closed.