Patient Experience Data Project Patient Engagement For Medicines

Patientengagement Patientexperiencedata Patientengagement Pfmd A dynamic tool, building on the global ped navigator, that illustrates how and when patients can be engaged in the generation and use of patient experience data. Working with patients through meaningful patient engagement (pe) and incorporating patient experience data (pxd) is increasingly important in medicines and medical device development.

Pfmd Org On Linkedin Patient Experience Data Patient Engagement For This reflection paper encourages stakeholders and developers to engage at an early stage with regulators to discuss inclusion of patient experience data (ped) in medicine development programmes and marketing authorisation applications (maas). Patient engagement in the development and regulation of medicines is an increasingly important element that aims to incorporate the views of patients throughout medicines’ life cycle, from research and development to regulatory approval and post marketing surveillance. We conducted a global review of the pe and pxd landscape to identify evolving resources by geography to support and highlight the potential of integration of pe and pxd in regulatory. Looking for a step by step guide to plan, access and execute the quality of your patient engagement activities in medicines development or medtech projects? the patient engagement quality guidance is your go to.

Patient Engagement And Patient Experience Data Understanding The Use We conducted a global review of the pe and pxd landscape to identify evolving resources by geography to support and highlight the potential of integration of pe and pxd in regulatory. Looking for a step by step guide to plan, access and execute the quality of your patient engagement activities in medicines development or medtech projects? the patient engagement quality guidance is your go to. To better understand what matters most to patients, patient experience data (ped) plays a critical role in the drug development lifecycle, including considering real world data (rwd) after launch. The european medicines agency (ema) has released a new draft reflection paper encouraging developers and other stakeholders to plan, generate, and submit patient experience data (ped) across the medicine lifecycle, and to engage early with regulators on methods and use cases. The european medicines agency (ema) has published a new reflection paper on the use of patient experience data (ped) in drug development, regulatory evaluation, and post market surveillance. Key elements for patient engagement in the development and life cycle management of medicines were identified. an overarching roadmap for a sustainable partnership among stakeholders is proposed.
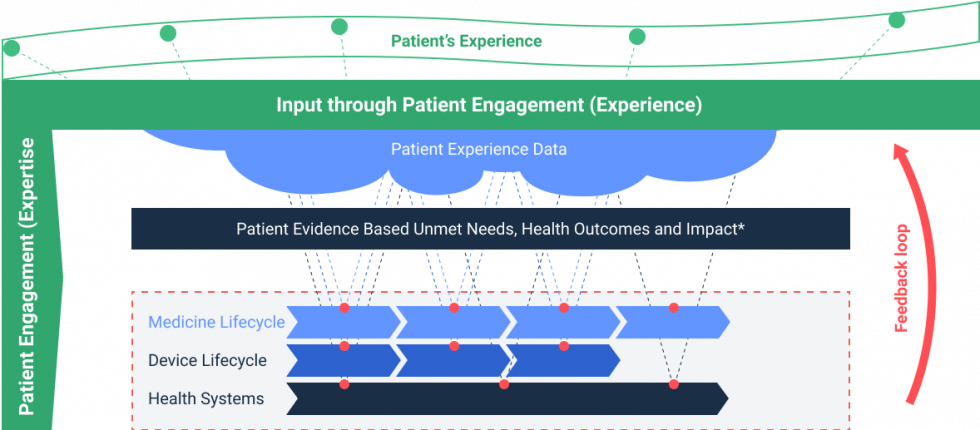
Patient Experience Data Project Patient Engagement For Medicines To better understand what matters most to patients, patient experience data (ped) plays a critical role in the drug development lifecycle, including considering real world data (rwd) after launch. The european medicines agency (ema) has released a new draft reflection paper encouraging developers and other stakeholders to plan, generate, and submit patient experience data (ped) across the medicine lifecycle, and to engage early with regulators on methods and use cases. The european medicines agency (ema) has published a new reflection paper on the use of patient experience data (ped) in drug development, regulatory evaluation, and post market surveillance. Key elements for patient engagement in the development and life cycle management of medicines were identified. an overarching roadmap for a sustainable partnership among stakeholders is proposed.
Comments are closed.