Oxidizing And Reducing Agents

Oxidizing And Reducing Agents Pdf Redox Chemical Reactions This page discusses what defines an oxidizing or reducing agent, how to determine an oxidizing and reducing agent in a chemical reaction, and the importance of this concept in real world applications. Learn how to identify the oxidizing and reducing agents in redox reactions using oxidation numbers and examples. find out the common oxidizing agents, the correlation between oxidation and reduction, and the exceptions and rules for determining oxidation states.
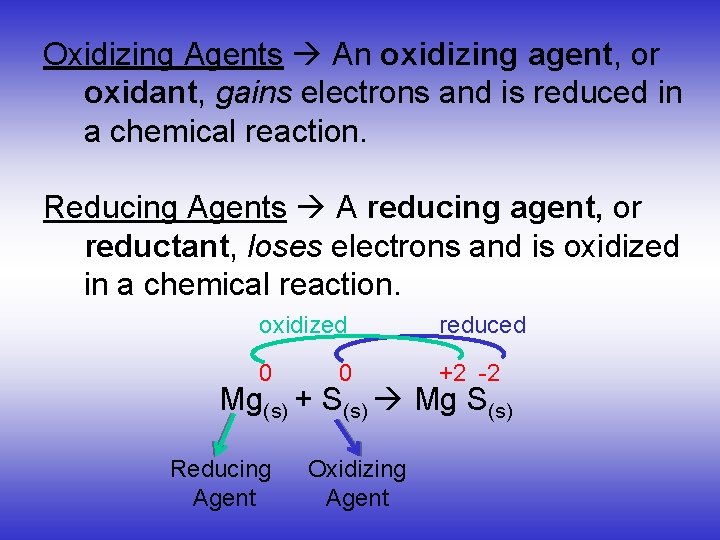
Oxidizing Reducing Agents Oxidizing Agents An Oxidizing Agent Learn about the most common oxidizing and reducing agents used in chemistry labs and experiments, with examples and infographics. find out how to handle them safely and what reactions they can cause. Oxidation is the loss of electron (s), and reduction is the gain of electron (s). one reactant, called the reducing agent or reductant, gives electron (s) to another reactant, called the oxidizing agent or oxidant. This tutorial covers oxidation reduction (redox) reactions in electrochemistry: learn to write simple half reactions and to recognize the oxidizing agent and reducing agent. Learn how to identify and classify oxidizing and reducing agents in chemical reactions. see examples, common oxidizing agents and reducing agents, and the relative strengths of oxidizing and reducing agents.

Oxidizing And Reducing Agents Chemistry Steps This tutorial covers oxidation reduction (redox) reactions in electrochemistry: learn to write simple half reactions and to recognize the oxidizing agent and reducing agent. Learn how to identify and classify oxidizing and reducing agents in chemical reactions. see examples, common oxidizing agents and reducing agents, and the relative strengths of oxidizing and reducing agents. Oxidants accept electrons, becoming reduced, while reductants donate electrons, becoming oxidized. strong oxidants react vigorously, while weaker ones are less reactive. Now, before we assign oxidizing and reducing agents, let's just go ahead and talk about this one more time, except showing all of the valence electrons. so let's also assign some oxidation states using this way because there are two ways to assign oxidation states. The reducing agent is a substance that causes reduction by losing electrons. the simplest way to think of this is that the oxidizing agent is the substance that is reduced, while the reducing agent is the substance that is oxidized. Examples of oxidizing agents include halogens, potassium nitrate, and nitric acid. a reducing agent, or reductant, loses electrons and is oxidized in a chemical reaction. a reducing agent typically is in one of its lower possible oxidation states and is known as the electron donor.
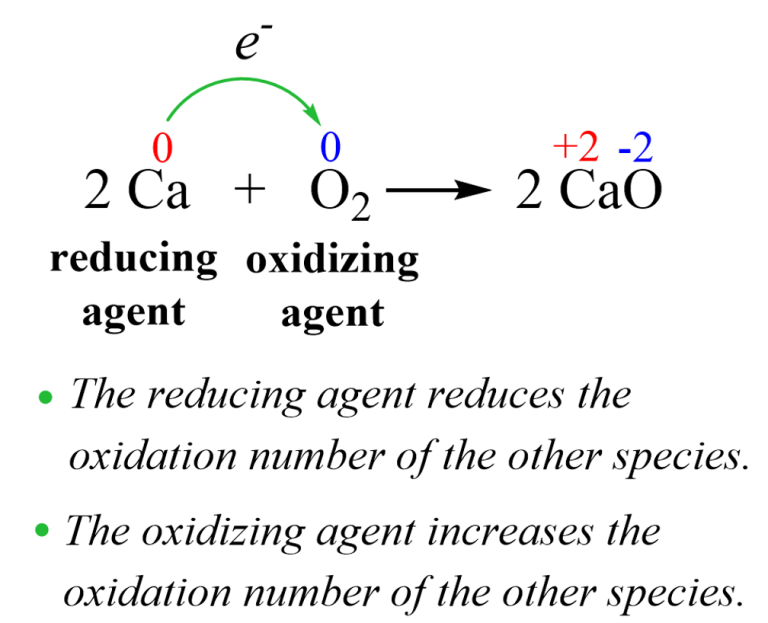
Oxidizing And Reducing Agents Chemistry Steps Oxidants accept electrons, becoming reduced, while reductants donate electrons, becoming oxidized. strong oxidants react vigorously, while weaker ones are less reactive. Now, before we assign oxidizing and reducing agents, let's just go ahead and talk about this one more time, except showing all of the valence electrons. so let's also assign some oxidation states using this way because there are two ways to assign oxidation states. The reducing agent is a substance that causes reduction by losing electrons. the simplest way to think of this is that the oxidizing agent is the substance that is reduced, while the reducing agent is the substance that is oxidized. Examples of oxidizing agents include halogens, potassium nitrate, and nitric acid. a reducing agent, or reductant, loses electrons and is oxidized in a chemical reaction. a reducing agent typically is in one of its lower possible oxidation states and is known as the electron donor.

Oxidizing And Reducing Agents Definition Examples Expii The reducing agent is a substance that causes reduction by losing electrons. the simplest way to think of this is that the oxidizing agent is the substance that is reduced, while the reducing agent is the substance that is oxidized. Examples of oxidizing agents include halogens, potassium nitrate, and nitric acid. a reducing agent, or reductant, loses electrons and is oxidized in a chemical reaction. a reducing agent typically is in one of its lower possible oxidation states and is known as the electron donor.
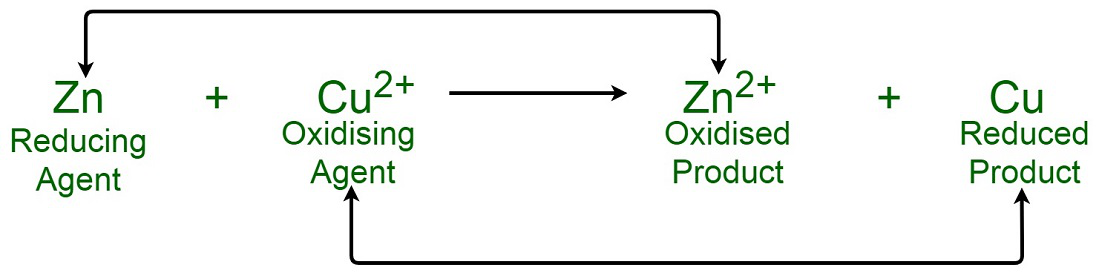
Oxidizing And Reducing Agents Geeksforgeeks
Comments are closed.