Oxidation And Reduction Fundamentals Of Redox Reactions
Introduction To Oxidation Reduction Redox Reactions Oxidation This tutorial covers oxidation reduction (redox) reactions in electrochemistry: learn to write simple half reactions and to recognize the oxidizing agent and reducing agent. Redox reactions involve the transfer of electrons between reactant species and are distinct from other reaction types, such as acid base, which involve the transfer of protons. oxidation is the loss of electron (s), and reduction is the gain of electron (s).

Oxidation Vs Reduction Mastering Redox Reactions Examples Using the oxidation number concept, an all inclusive definition of redox reaction has been established. oxidation reduction (redox) reactions are those in which one or more elements involved undergo a change in oxidation number. An oxidation reduction (redox) reaction is a type of chemical reaction that involves a transfer of electrons between two species. an oxidation reduction reaction is any chemical reaction in which the oxidation number of a molecule, atom, or ion changes by gaining or losing an electron. Learn what a redox reaction is and how to identify and balance redox reactions. explore oxidation, reduction, and oxidation numbers. This tutorial covers oxidation, reduction, how the two combine to form redox reactions and the types of redox reactions and examples.

Introduction To Oxidation Reduction And Redox Reactions Redox I Learn what a redox reaction is and how to identify and balance redox reactions. explore oxidation, reduction, and oxidation numbers. This tutorial covers oxidation, reduction, how the two combine to form redox reactions and the types of redox reactions and examples. Redox processes are defined as reactions accompanied by oxidation state changes: an increase in an atom’s oxidation number corresponds to an oxidation; a decrease, to a reduction. Reactions in which electrons are transferred, resulting in oxidation and reduction, are called oxidation reduction reactions. because the term oxidation reduction is a bit cumbersome, we usually call these reactions redox reactions. Oxidation and reduction reactions, commonly referred to as redox reactions, are truly ubiquitous in chemistry, underpinning processes in virtually every branch, from foundational inorganic and organic chemistry to intricate biochemical pathways and large scale industrial applications. This is the 'ordinary molecular' equation for a typical metal displacement reaction, but this does not really show what happens in terms of atoms, ions and electrons, so we use ionic equations like the one shown below.
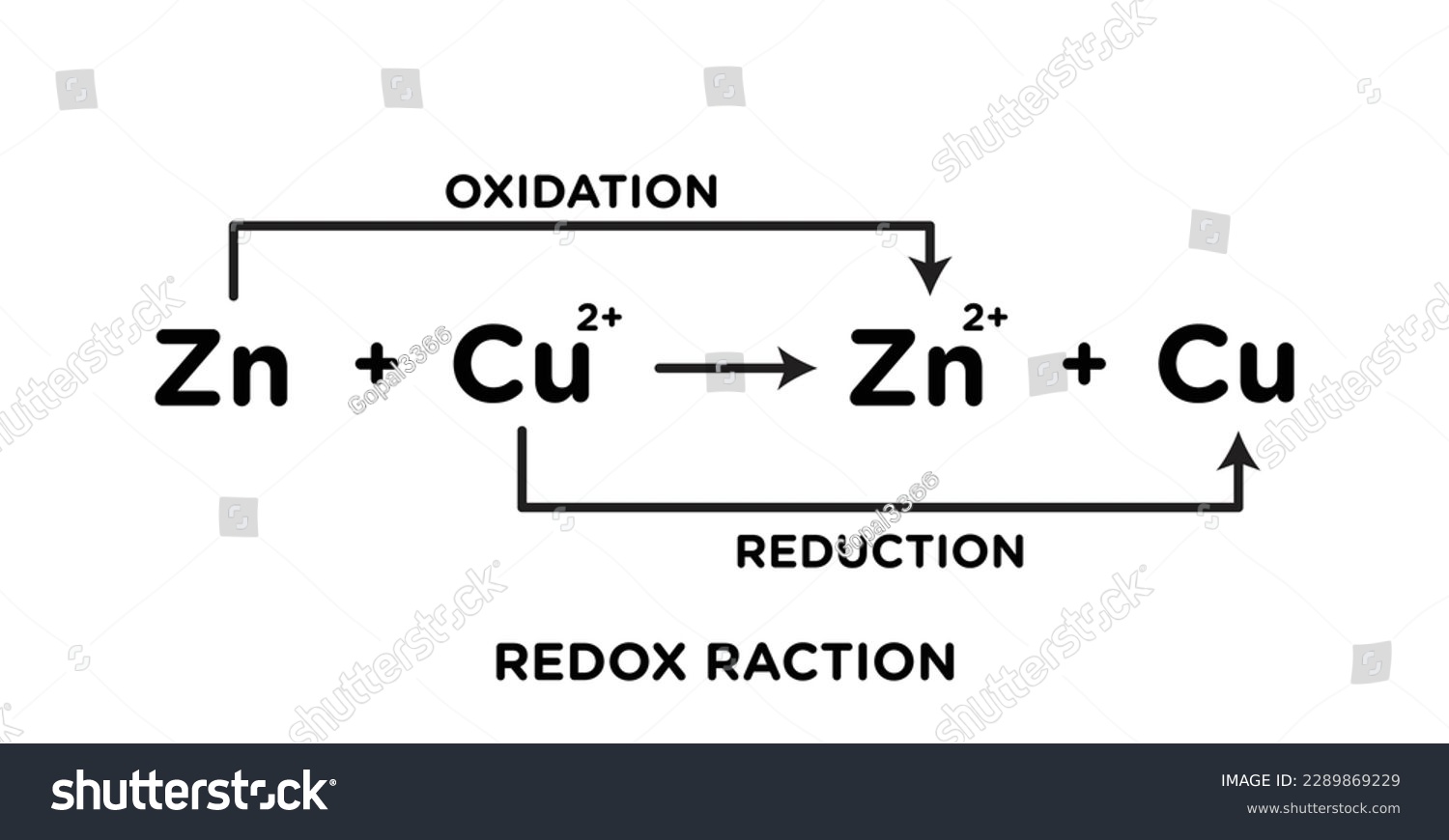
Redox Reaction Oxidation Reduction Reactions Stock Vector Royalty Free Redox processes are defined as reactions accompanied by oxidation state changes: an increase in an atom’s oxidation number corresponds to an oxidation; a decrease, to a reduction. Reactions in which electrons are transferred, resulting in oxidation and reduction, are called oxidation reduction reactions. because the term oxidation reduction is a bit cumbersome, we usually call these reactions redox reactions. Oxidation and reduction reactions, commonly referred to as redox reactions, are truly ubiquitous in chemistry, underpinning processes in virtually every branch, from foundational inorganic and organic chemistry to intricate biochemical pathways and large scale industrial applications. This is the 'ordinary molecular' equation for a typical metal displacement reaction, but this does not really show what happens in terms of atoms, ions and electrons, so we use ionic equations like the one shown below.

Introduction To Oxidation Reduction Redox Reactions Oxidation and reduction reactions, commonly referred to as redox reactions, are truly ubiquitous in chemistry, underpinning processes in virtually every branch, from foundational inorganic and organic chemistry to intricate biochemical pathways and large scale industrial applications. This is the 'ordinary molecular' equation for a typical metal displacement reaction, but this does not really show what happens in terms of atoms, ions and electrons, so we use ionic equations like the one shown below.
Comments are closed.