Obtaining Informed Consent

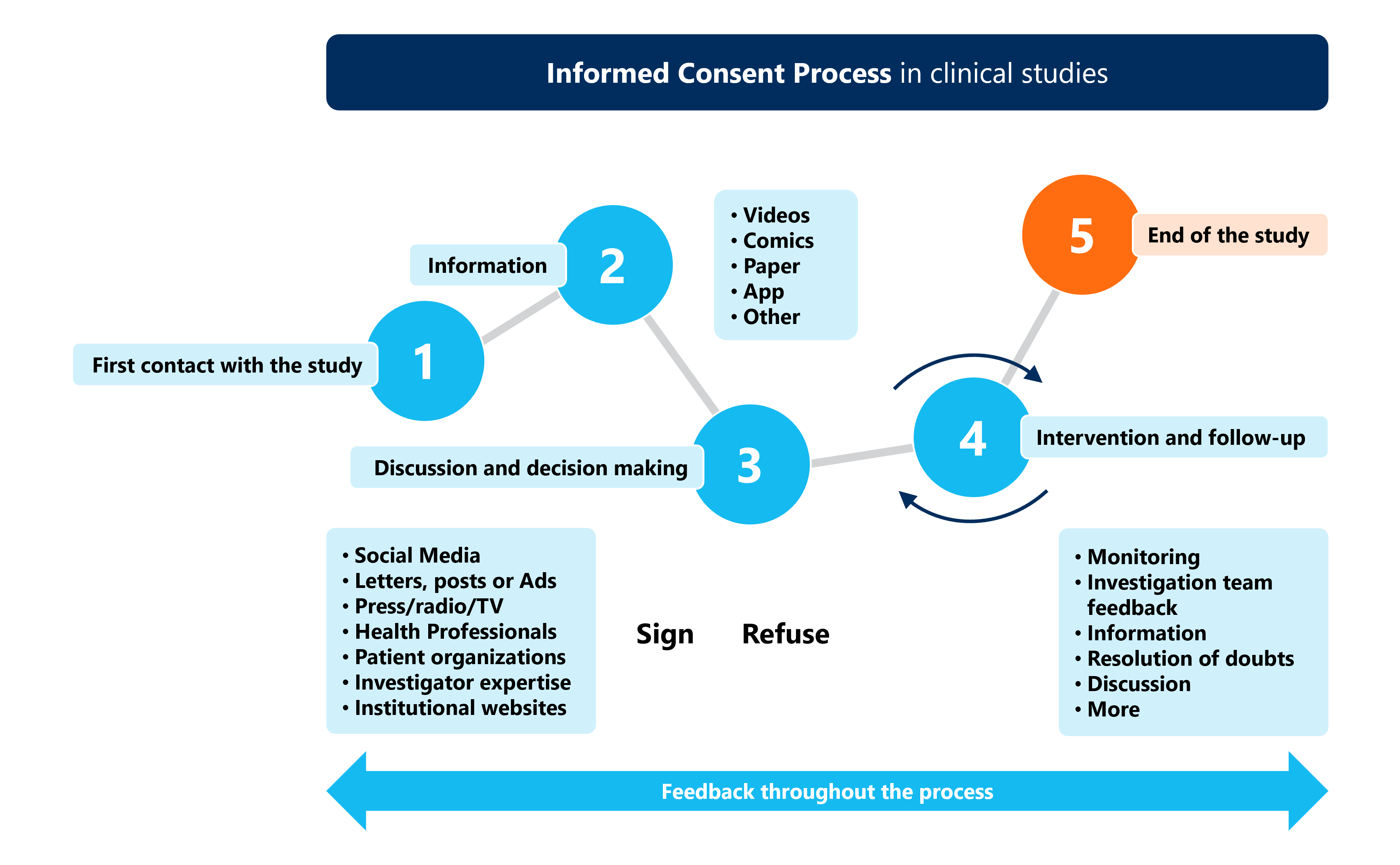
Graphical Model Of Obtaining Informed Consent Form Process Download The informed consent process should ensure that all critical information about a study is completely disclosed, and that prospective subjects or their legally authorized representatives adequately understand the research so that they can make informed choices. Obtaining consent involves explaining the research and assessing participant comprehension using a consent document, usually a written consent form or information sheet, as a guide for the verbal explanation of the study.

Graphical Model Of Obtaining Informed Consent Form Process Download Informed consent process is the cornerstone of ethics in clinical research. obtaining informed consent from patients participating in clinical research is an important legal and ethical imperative for clinical trial researchers. Obtaining genuine informed consent from research participants is best thought of as a process of sharing information and addressing questions and concerns, rather than simply obtaining a signature on a prescribed form. This paper provides a comprehensive review of the tenets underlying informed consent in research, including the assessment of capacity to consent, considerations for patients unable to consent, when to seek consent from substitute decision makers, and consent under special circumstances. What does it mean to give my informed consent? when you give your informed consent, you say it’s ok for your healthcare provider to move forward with a certain treatment or test.

How To Obtain Informed Consent From Research Participants This paper provides a comprehensive review of the tenets underlying informed consent in research, including the assessment of capacity to consent, considerations for patients unable to consent, when to seek consent from substitute decision makers, and consent under special circumstances. What does it mean to give my informed consent? when you give your informed consent, you say it’s ok for your healthcare provider to move forward with a certain treatment or test. Obtaining consent involves informing the subject about his or her rights, the purpose of the study, the procedures to be undergone, and the potential risks and benefits of their participation. Learn the core of informed consent in clinical research. build trust, ensure transparency, and safeguard participant rights with expert insights. What is informed consent? investigators must educate potential participants to ensure that they can reach a truly informed decision about whether to participate in the research. informed consent must be given freely, without coercion, and must be based on a clear understanding of what participation involves interactive discussion. Always refer to the overseeing irb, and local (institutional) requirements to determine applicable policies or guidance for obtaining and documenting informed consent. always follow the consent process, as detailed in irb approved protocol.

Q A How To Obtain Informed Consent From Medical Research Participants Obtaining consent involves informing the subject about his or her rights, the purpose of the study, the procedures to be undergone, and the potential risks and benefits of their participation. Learn the core of informed consent in clinical research. build trust, ensure transparency, and safeguard participant rights with expert insights. What is informed consent? investigators must educate potential participants to ensure that they can reach a truly informed decision about whether to participate in the research. informed consent must be given freely, without coercion, and must be based on a clear understanding of what participation involves interactive discussion. Always refer to the overseeing irb, and local (institutional) requirements to determine applicable policies or guidance for obtaining and documenting informed consent. always follow the consent process, as detailed in irb approved protocol.
Informed Consent Comic What is informed consent? investigators must educate potential participants to ensure that they can reach a truly informed decision about whether to participate in the research. informed consent must be given freely, without coercion, and must be based on a clear understanding of what participation involves interactive discussion. Always refer to the overseeing irb, and local (institutional) requirements to determine applicable policies or guidance for obtaining and documenting informed consent. always follow the consent process, as detailed in irb approved protocol.
Comments are closed.