Nomenclature Of Acids Pathways To Chemistry
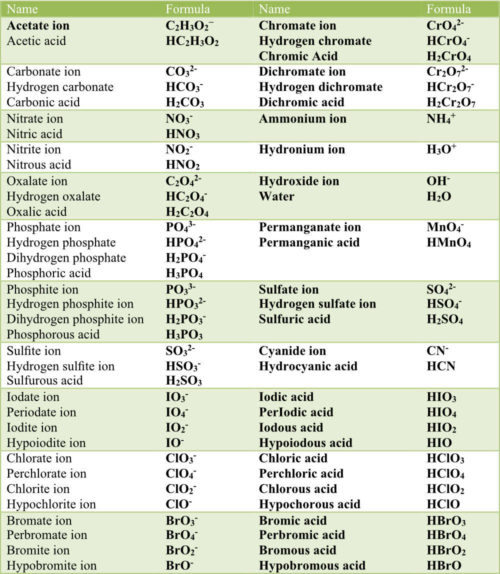
Polyatomicionsandacids Pathways To Chemistry In this study guide we will learn how to name acids and bases. first we will start with acids. the table below lists some common polyatomic ions. it is assumed you have already memorized the names, formulas, and charges of the polyatomic ions. oxoacids are acids that contain oxygen atoms. This document covers nomenclature, structure, and properties of carboxylic acids and their derivatives, including iupac naming, boiling points, acidity, and preparation reactions. it includes various questions and answers to assess understanding of these concepts.
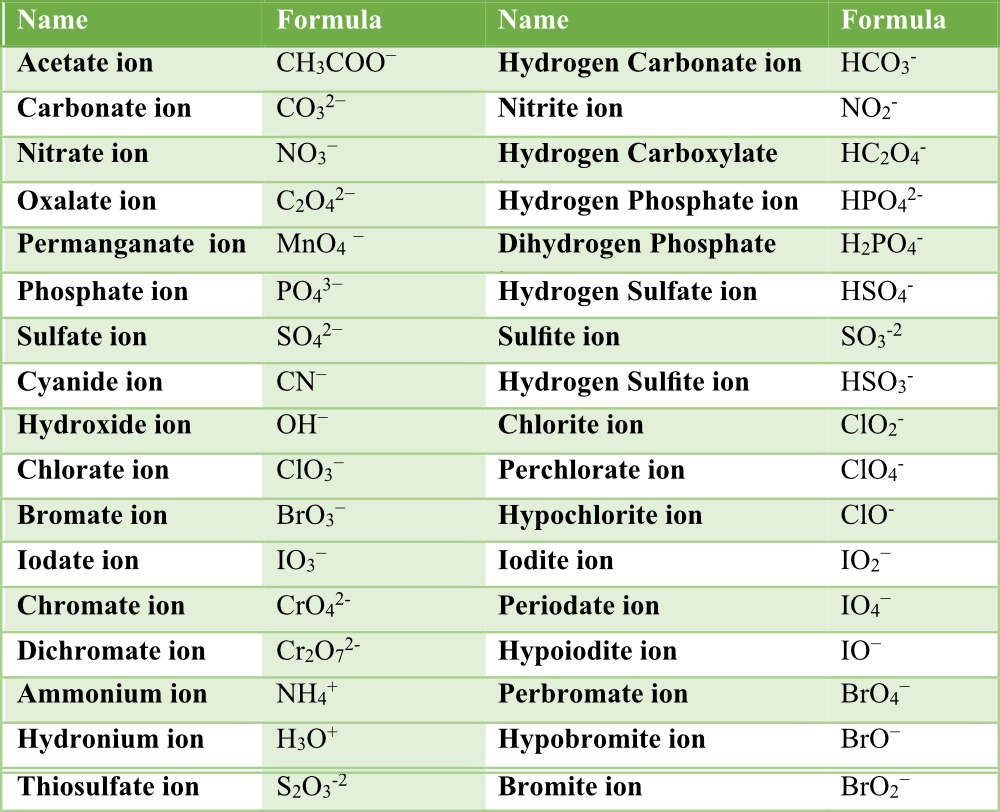
Nomenclature Of Acids Pathways To Chemistry Nomenclature answer key back to worksheets back to nomenclature study guide back to naming binary covalent compounds study guide back to naming simple ionic compounds study guide back to ionic compounds with polyatomic ions study guide back to nomenclature of acids study guide. Write chemical formulas for the following acids. back to nomenclature of acids. solutions to exercises exercise 1. name the following acids. 1. h2so3; sulfurous acid 2. h2te; hydrotelluric acid 3. h2cro4; chromic acid 4. hclo2; chlorous acid 5. ch3cooh; acetic acid exercise 2. write chemical formulas for the following acids. 1. In these worksheets, students will dive deep into the rules and conventions for naming various acids, from the familiar ones found in everyday substances to more complex compounds encountered in advanced chemistry. Some compounds containing hydrogen are members of an important class of substances known as acids. the chemistry of these compounds is explored in more detail in later chapters of this text, but for now, it will suffice to note that many acids release hydrogen ions, h , when dissolved in water.

Nomenclature Of Acids Pathways To Chemistry In these worksheets, students will dive deep into the rules and conventions for naming various acids, from the familiar ones found in everyday substances to more complex compounds encountered in advanced chemistry. Some compounds containing hydrogen are members of an important class of substances known as acids. the chemistry of these compounds is explored in more detail in later chapters of this text, but for now, it will suffice to note that many acids release hydrogen ions, h , when dissolved in water. As you become more sophisticated in your chemistry, you will realize that there are many acid formulas that do not start with h, but those will almost all be left for another time. Si [email protected] 9.11.2025 chem 223 – si sheet #4 material: chapter 2 and 3 acids review, iupac nomenclature session objectives. upon completion of this session, participants will be able to compare acid and base strengths, acidity basicity based on pka identify nucleophiles and electrophiles in acid base reactions predict possible products and reaction mechanisms in acid base. Acids are molecular compounds that release hydrogen ions. a binary acid consists of hydrogen and one other element. oxyacids contain hydrogen, oxygen, and one other element. the name of the acid is based on the anion attached to the hydrogen. But to identify polyatomic ions derived from acids as a single unit, you must necessarily know the polyatomic acid from whence the anion came. type i metal — denotes a metal with only one oxidation number. that is, upon ionization, the metal will always produce an ion of the same charge.

Nomenclature Of Carboxylic Acids Organic Chemistry Tutor As you become more sophisticated in your chemistry, you will realize that there are many acid formulas that do not start with h, but those will almost all be left for another time. Si [email protected] 9.11.2025 chem 223 – si sheet #4 material: chapter 2 and 3 acids review, iupac nomenclature session objectives. upon completion of this session, participants will be able to compare acid and base strengths, acidity basicity based on pka identify nucleophiles and electrophiles in acid base reactions predict possible products and reaction mechanisms in acid base. Acids are molecular compounds that release hydrogen ions. a binary acid consists of hydrogen and one other element. oxyacids contain hydrogen, oxygen, and one other element. the name of the acid is based on the anion attached to the hydrogen. But to identify polyatomic ions derived from acids as a single unit, you must necessarily know the polyatomic acid from whence the anion came. type i metal — denotes a metal with only one oxidation number. that is, upon ionization, the metal will always produce an ion of the same charge.
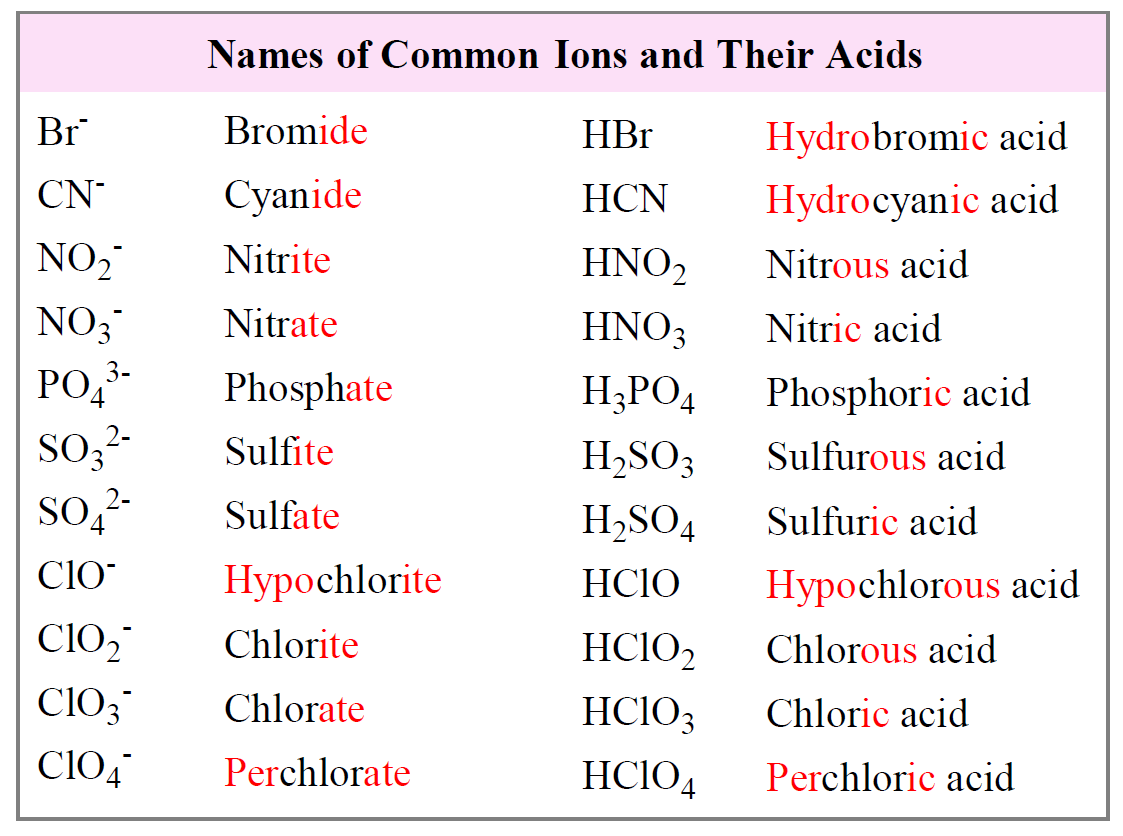
Naming Acids And Bases Chemistry Steps Acids are molecular compounds that release hydrogen ions. a binary acid consists of hydrogen and one other element. oxyacids contain hydrogen, oxygen, and one other element. the name of the acid is based on the anion attached to the hydrogen. But to identify polyatomic ions derived from acids as a single unit, you must necessarily know the polyatomic acid from whence the anion came. type i metal — denotes a metal with only one oxidation number. that is, upon ionization, the metal will always produce an ion of the same charge.

Nomenclature Acids Chem 9 Studocu
Comments are closed.