Naming Acids Simplified

Key Naming Acids Pdf Learn what acids are, what classifies an acid as strong or weak, and the acid naming rules! step by step instructions with examples on acid naming rules. Learn how to name acids in chemistry. why does it matter. explore easy rules, examples, and charts for binary acids and oxyacids.
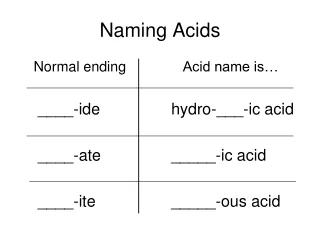
Ppt Naming Acids Powerpoint Presentation Free Download Id 1951386 Acids have their own nomenclature system. if an acid is composed of only hydrogen and one other element, the name is hydro the stem of the other element ic acid. for example, the compound hcl (aq) is hydrochloric acid, while h 2 s (aq) is hydrosulfuric acid. You can still think of an acid as an ionic compound, with the hydrogen ion (h a ) acting as the metal cation. but an acid gets a special name based on the anion that follows the hydrogen. The most straightforward definition is that an acid is a molecular compound that contains one or more hydrogen atoms and produces hydrogen ions (h ) when dissolved in water. Learn the simple steps to correctly name acids, including binary acids and oxyacids. this guide breaks down the process into easy to follow instructions, helping you master acid nomenclature with confidence. perfect for chemistry students and enthusiasts looking to deepen their understanding.
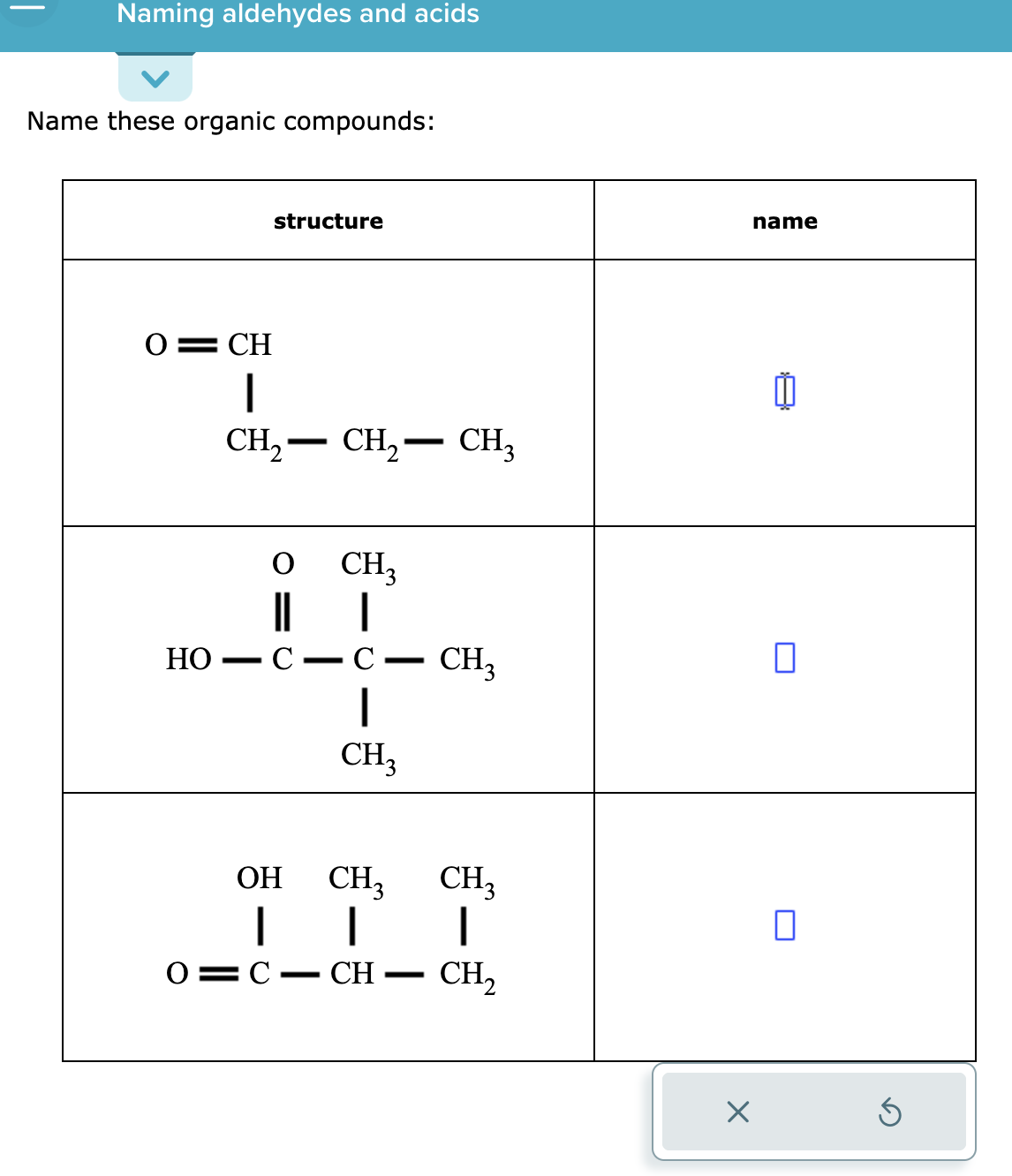
Solved Naming Aldehydes And Acids Name These Organic Chegg The most straightforward definition is that an acid is a molecular compound that contains one or more hydrogen atoms and produces hydrogen ions (h ) when dissolved in water. Learn the simple steps to correctly name acids, including binary acids and oxyacids. this guide breaks down the process into easy to follow instructions, helping you master acid nomenclature with confidence. perfect for chemistry students and enthusiasts looking to deepen their understanding. To name an acid, first determine if it contains oxygen. for acids without oxygen (binary acids), use the prefix "hydro " and change the anion's ending to " ic acid" (e.g., hcl becomes hydrochloric acid). Acids, broadly defined as substances that donate protons (h ) or accept electrons, are classified into two primary categories: binary acids and oxoacids. the naming convention differs substantially between these categories. Initially, we will define an acid as an ionic compound of the h cation dissolved in water. to indicate that something is dissolved in water, we will use the phase label (aq) next to a chemical formula (where aq stands for “aqueous,” a word that describes something dissolved in water). The name of acid is derived from the name of its anion. each ending and prefix of an anion forms a specific combination in the name of the acid.

Naming Acids To name an acid, first determine if it contains oxygen. for acids without oxygen (binary acids), use the prefix "hydro " and change the anion's ending to " ic acid" (e.g., hcl becomes hydrochloric acid). Acids, broadly defined as substances that donate protons (h ) or accept electrons, are classified into two primary categories: binary acids and oxoacids. the naming convention differs substantially between these categories. Initially, we will define an acid as an ionic compound of the h cation dissolved in water. to indicate that something is dissolved in water, we will use the phase label (aq) next to a chemical formula (where aq stands for “aqueous,” a word that describes something dissolved in water). The name of acid is derived from the name of its anion. each ending and prefix of an anion forms a specific combination in the name of the acid.

Acids Naming Quiz Quiz Now Initially, we will define an acid as an ionic compound of the h cation dissolved in water. to indicate that something is dissolved in water, we will use the phase label (aq) next to a chemical formula (where aq stands for “aqueous,” a word that describes something dissolved in water). The name of acid is derived from the name of its anion. each ending and prefix of an anion forms a specific combination in the name of the acid.
Comments are closed.