Naming Acids Pdf

Naming Acids Pdf Naming oxyacids: these are more difficult to name because these acids have hydrogen, a nonmetal, and may have varying numbers of oxygen atoms. for example, h2so5, h2so4, h2so3, and h2so2 are all acids. Binary acids are formed when hydrogen combines with another non metal. acids are dissolved water, so therefore they are always followed by the symbol aqueous or ‘aq.’.

Naming Acids Worksheet Naming acids free download as pdf file (.pdf) or read online for free. the document explains the unique characteristics of acids and their naming conventions, distinguishing between binary and ternary acids. Some compounds containing hydrogen are members of an important class of substances known as acids. the chemistry of these compounds is explored in more detail in later chapters of this text, but for now, it will suffice to note that many acids release hydrogen ions, h , when dissolved in water. Follow these rules: determine the name of the polyatomic ion. d –ite ending to –ous. keep hypo and per prefixes. You should be able to write the names of the following binary acids given the formula, or write the formula given the name. memorize the names and formulas of the following acids:.

Naming Acids And Bases Online Exercise For Worksheets Library Follow these rules: determine the name of the polyatomic ion. d –ite ending to –ous. keep hypo and per prefixes. You should be able to write the names of the following binary acids given the formula, or write the formula given the name. memorize the names and formulas of the following acids:. The table below includes both binary and ternary acids. using what you have learned in this activity, fill in the missing formula and name for the anion in each acid, and give the formula of the acid. How to name acids is a fundamental topic in chemistry that bridges the gap between molecular structure and communication within scientific communities. accurate naming conventions enable chemists, educators, and students to convey complex chemical information succinctly and universally. 5 ternary acids contain h, o, and a nonmetal. name the following ternary acids: (review the polyatomic ions nomenclature if you feel you have forgotten a few!). An acid can be defined in several ways. the most straightforward definition is that an acid is a molecular compound that contains one or more hydrogen atoms and produces hydrogen ions (h a ) when dissolved in water.
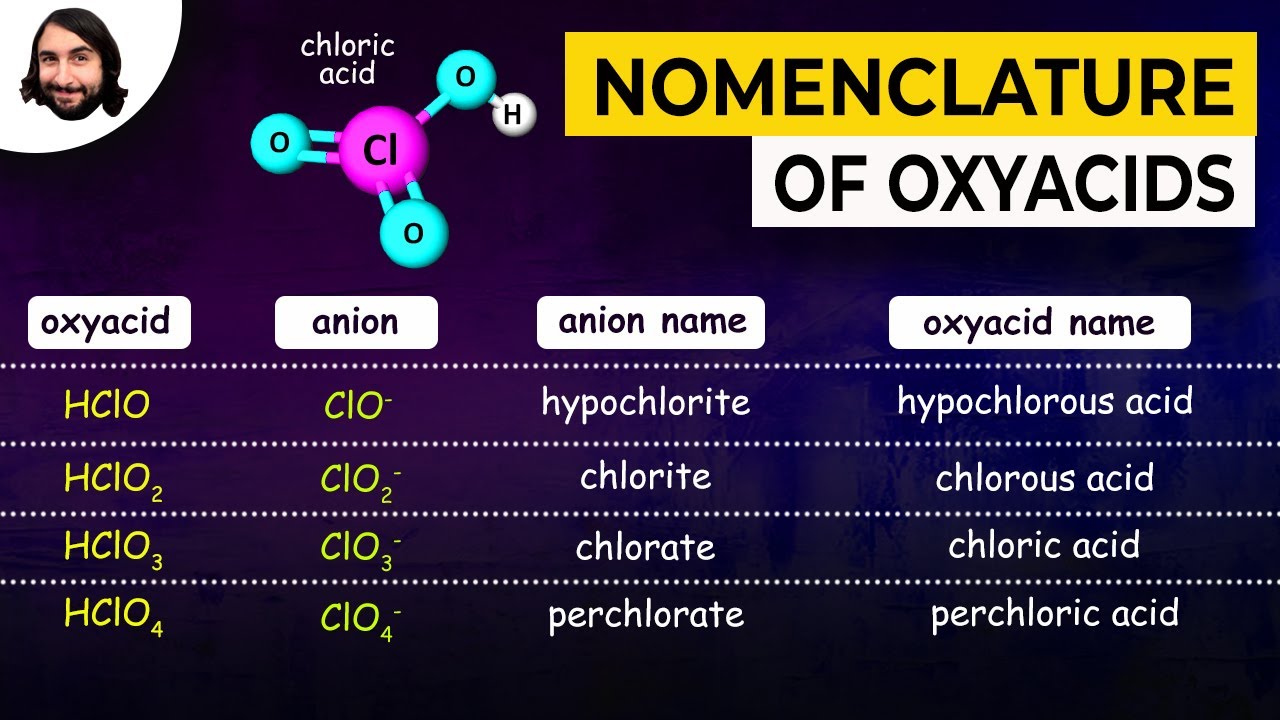
Naming Acids Worksheet Hno3 The table below includes both binary and ternary acids. using what you have learned in this activity, fill in the missing formula and name for the anion in each acid, and give the formula of the acid. How to name acids is a fundamental topic in chemistry that bridges the gap between molecular structure and communication within scientific communities. accurate naming conventions enable chemists, educators, and students to convey complex chemical information succinctly and universally. 5 ternary acids contain h, o, and a nonmetal. name the following ternary acids: (review the polyatomic ions nomenclature if you feel you have forgotten a few!). An acid can be defined in several ways. the most straightforward definition is that an acid is a molecular compound that contains one or more hydrogen atoms and produces hydrogen ions (h a ) when dissolved in water.
Comments are closed.