Naming Acids Introduction

Naming Acids Pdf Learn how to name acids in chemistry. why does it matter. explore easy rules, examples, and charts for binary acids and oxyacids. We'll learn how to look at the chemical formula for an acid and then write its name. we will focus on both acids without oxygen and also acids that contain oxygen, which are sometimes.

Key Naming Acids Pdf Initially, we will define an acid as an ionic compound of the h cation dissolved in water. to indicate that something is dissolved in water, we will use the phase label (aq) next to a chemical formula (where aq stands for “aqueous,” a word that describes something dissolved in water). This page discusses the spot test for gold in the context of the california gold rush, explaining both the definition and properties of acids. it details how acids release hydrogen ions in water and …. How do you name acids? we'll learn how to look at the chemical formula for an acid and then write its name. Understanding the basic structure of acids sets the foundation for exploring the various types of acids and the rules for naming them, which will be discussed further in subsequent lessons.

Naming Acids Packet Pdf Acid Sulfuric Acid How do you name acids? we'll learn how to look at the chemical formula for an acid and then write its name. Understanding the basic structure of acids sets the foundation for exploring the various types of acids and the rules for naming them, which will be discussed further in subsequent lessons. The name of acid is derived from the name of its anion. each ending and prefix of an anion forms a specific combination in the name of the acid. You can still think of an acid as an ionic compound, with the hydrogen ion (h a ) acting as the metal cation. but an acid gets a special name based on the anion that follows the hydrogen. Acids are named by the anion they form when dissolved in water. depending on what anion the hydrogen is attached to, acids will have different names. simple acids, known as binary acids, have only one anion and one hydrogen. these anions usually have the ending " ide.". Understanding the rules for naming acids is foundational to chemistry. an acid is typically defined as a substance that yields hydrogen ions (h ) when dissolved in water. the nomenclature, or naming system, depends entirely on whether the acid contains oxygen (an oxyacid) or not (a binary acid).
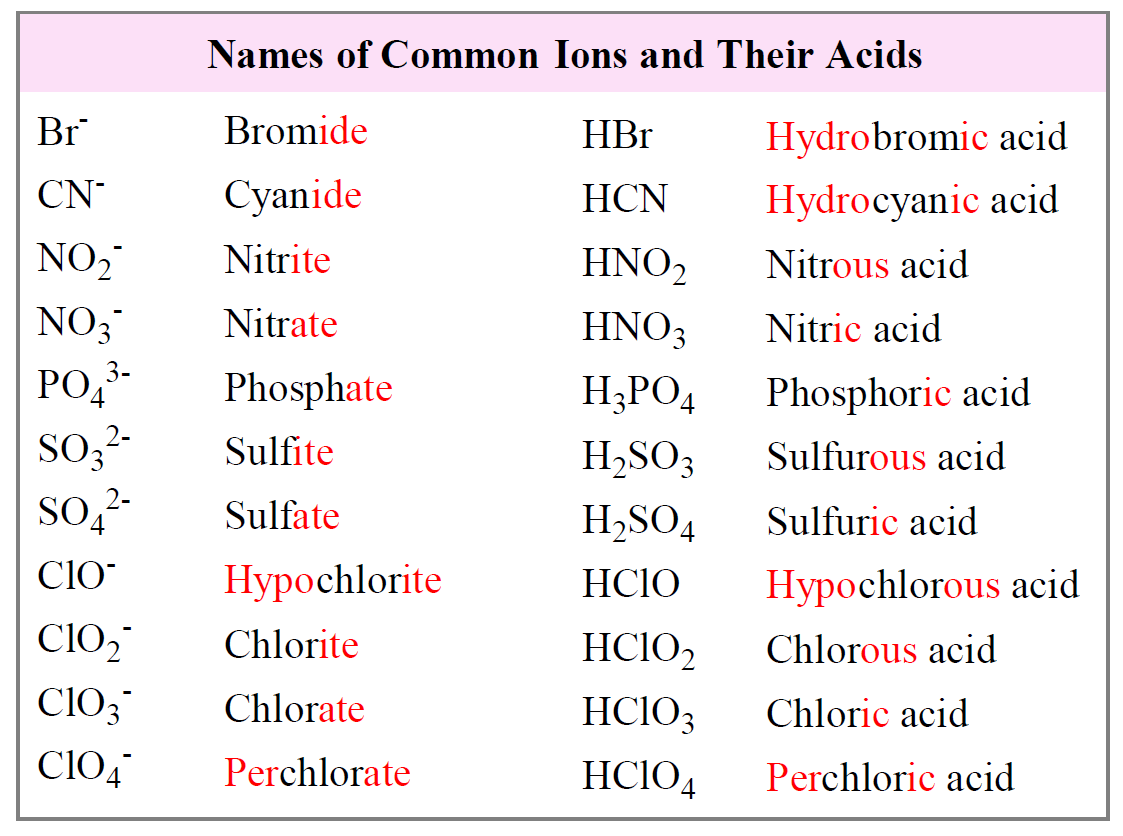
Naming Acids And Bases Chemistry Steps The name of acid is derived from the name of its anion. each ending and prefix of an anion forms a specific combination in the name of the acid. You can still think of an acid as an ionic compound, with the hydrogen ion (h a ) acting as the metal cation. but an acid gets a special name based on the anion that follows the hydrogen. Acids are named by the anion they form when dissolved in water. depending on what anion the hydrogen is attached to, acids will have different names. simple acids, known as binary acids, have only one anion and one hydrogen. these anions usually have the ending " ide.". Understanding the rules for naming acids is foundational to chemistry. an acid is typically defined as a substance that yields hydrogen ions (h ) when dissolved in water. the nomenclature, or naming system, depends entirely on whether the acid contains oxygen (an oxyacid) or not (a binary acid).
Comments are closed.