Named Thermodynamic Quantities

Thermodynamic Quantities Pdf Zirconium Metals Common thermodynamic equations and quantities in thermodynamics, using mathematical notation, are as follows:. Standard thermodynamic quantities for chemical substances at 25°c. source of data: crc handbook of chemistry and physics,84th edition (2004).

Table Of Thermodynamic Quantities Pdf Pdf Values of thermodynamic functions in the standard state characterized by a standard pressure, molality or amount concentration, but not by temperature. standard quantities are denoted by adding the superscript ⦵ ⦵ or ∘ to the symbol of the quantity. Explore thermodynamic values with this comprehensive table from the university of wisconsin–madison's general chemistry department. Semantic map subjects thermopedia fundamentals thermodynamics fluid flow basic concepts equations of motion non dimensional analysis euler number froude number knudsen number mach number quantities and units. Table of thermodynamic equations for more elaboration on these equations see: thermodynamic equations. the following page is a concise list of common thermodynamic equations and quantities:.
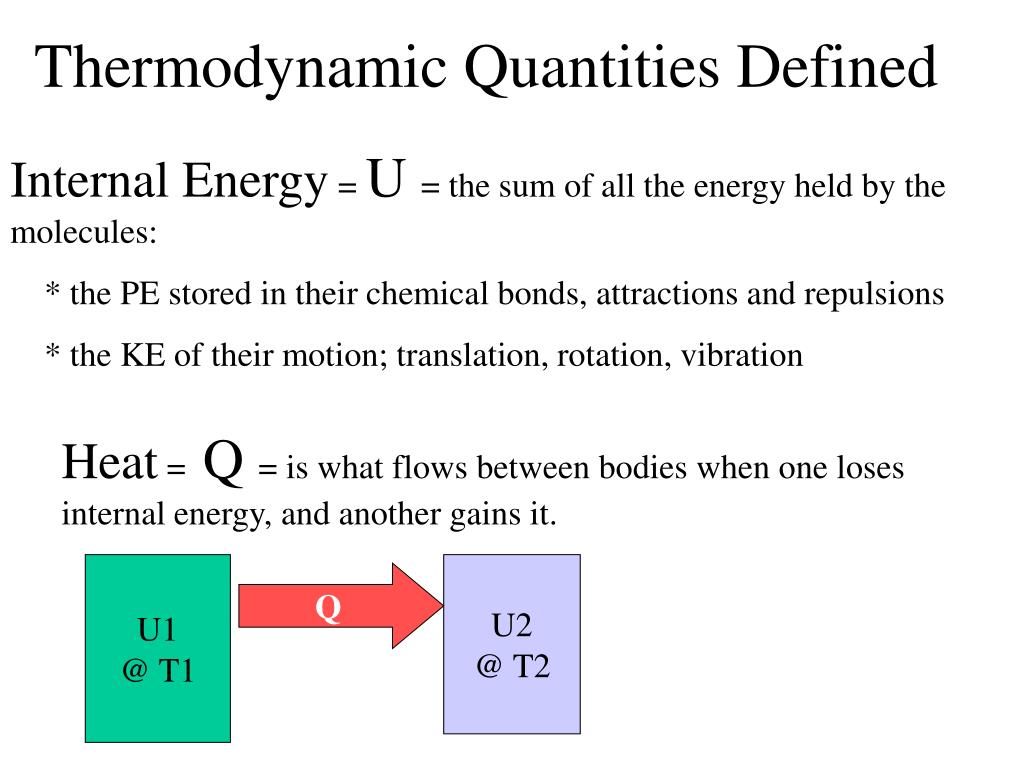
Ppt Thermodynamic Quantities Defined Powerpoint Presentation Free Semantic map subjects thermopedia fundamentals thermodynamics fluid flow basic concepts equations of motion non dimensional analysis euler number froude number knudsen number mach number quantities and units. Table of thermodynamic equations for more elaboration on these equations see: thermodynamic equations. the following page is a concise list of common thermodynamic equations and quantities:. Concise table of thermodynamic properties: heat, work, energy, entropy, with definitions, symbols, si units. ideal for chemistry students. The state of a thermodynamic system is specified by a number of extensive quantities, the most familiar of which are volume, internal energy, and the amount of each constituent particle (particle numbers). Synopsis. the standard chemical potential p is defined in terms of a specified state of aggregation, a specified temperature and a specified standard pressure. other standard thermodynamic quantities are obtained from p by the usual mathematical operations. It is traditional to divide thermodynamic quantities into two classes: “extensive” and “intensive”. extensive quantities increase linearly with system size while intensive quantities are independent of system size.
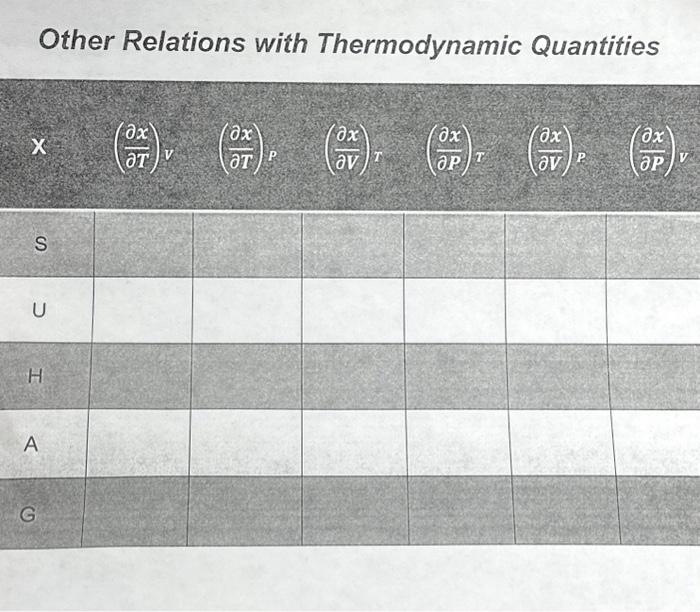
Other Relations With Thermodynamic Quantities Chegg Concise table of thermodynamic properties: heat, work, energy, entropy, with definitions, symbols, si units. ideal for chemistry students. The state of a thermodynamic system is specified by a number of extensive quantities, the most familiar of which are volume, internal energy, and the amount of each constituent particle (particle numbers). Synopsis. the standard chemical potential p is defined in terms of a specified state of aggregation, a specified temperature and a specified standard pressure. other standard thermodynamic quantities are obtained from p by the usual mathematical operations. It is traditional to divide thermodynamic quantities into two classes: “extensive” and “intensive”. extensive quantities increase linearly with system size while intensive quantities are independent of system size.
Comments are closed.