Mtr4

Rna Loading To Human Mtr4 Exosome Complex A Dna Rna Chimera Efficient, fast and flexible, the programmable mtr 4 transducer offers the same performance as up to four standard transducers, measuring and calculating ac voltage, ac current, active reactive apparent power, power factor, frequency, kwh, kvar, thd, dynamic and maximum demands. Mtr4 is a ski2 like rna helicase that plays a central role in rna surveillance and degradation pathways as an activator of the rna exosome. multiple crystallographic and cryo em studies over the past 10 years have revealed important insight into the.

The Nuclear Exosome Co Factor Mtr4 Shapes The Transcriptome For Meiotic Mtr4 is a cofactor of the nuclear rna exosome that degrades normal rnas and shapes the transcriptome for the transition from mitosis to meiosis in germ cells. mtr4 knockout mice are embryonic lethal and male infertile, and show defects in meiotic gene expression and alternative splicing. Mtr4 ensures the normal post transcriptional processing of maternal rnas, their nuclear export to the cytoplasm, and the accumulation of properly processed transcripts. oocytes with mtr4 knockout fail to accumulate sufficient and normal transcripts in the cytoplasm and cannot grow to normal sizes. In addition, act.d treatment decreased the nucleolar accumulation of the rna exosome by regulating nucleolar mtr4 levels. our work revealed that the subcellular localization of the rna exosome complex within the nucleolus can be regulated by mtr4. Transcriptomic analysis revealed a high expression of the methionine transporter slc1a5, which is regulated at the mrna level by the nuclear export factor mtr4. under conditions of methionine starvation, mtr4 undergoes methylation, leading to its ubiquitination.

Structural Basis For Mtr4 Zcchc8 Interactions That Stimulate The Mtr4 In addition, act.d treatment decreased the nucleolar accumulation of the rna exosome by regulating nucleolar mtr4 levels. our work revealed that the subcellular localization of the rna exosome complex within the nucleolus can be regulated by mtr4. Transcriptomic analysis revealed a high expression of the methionine transporter slc1a5, which is regulated at the mrna level by the nuclear export factor mtr4. under conditions of methionine starvation, mtr4 undergoes methylation, leading to its ubiquitination. Mtr4 ensures the normal post transcriptional processing of maternal rnas, their nuclear export to the cytoplasm, and the accumulation of properly processed transcripts. oocytes with mtr4 knockout fail to accumulate sufficient and normal transcripts in the cytoplasm and cannot grow to normal sizes. The most striking and unexpected feature of the mtr4 structure is a large, 265 residue domain that arches over the rest of the mtr4 structure and accounts for approximately 25% of the mtr4. The article explores how the human rna helicase mtr4 interacts with different rna binding proteins that function as adaptors for the nuclear exosome complex. it reveals the structural and functional diversity of the adaptors and their binding motifs on mtr4. The saccharomyces cerivisiae mrna transport protein (mtr4) is a rna helicase that interacts with the rna exosome. the exosome core is responsible for processing and degrading rna.
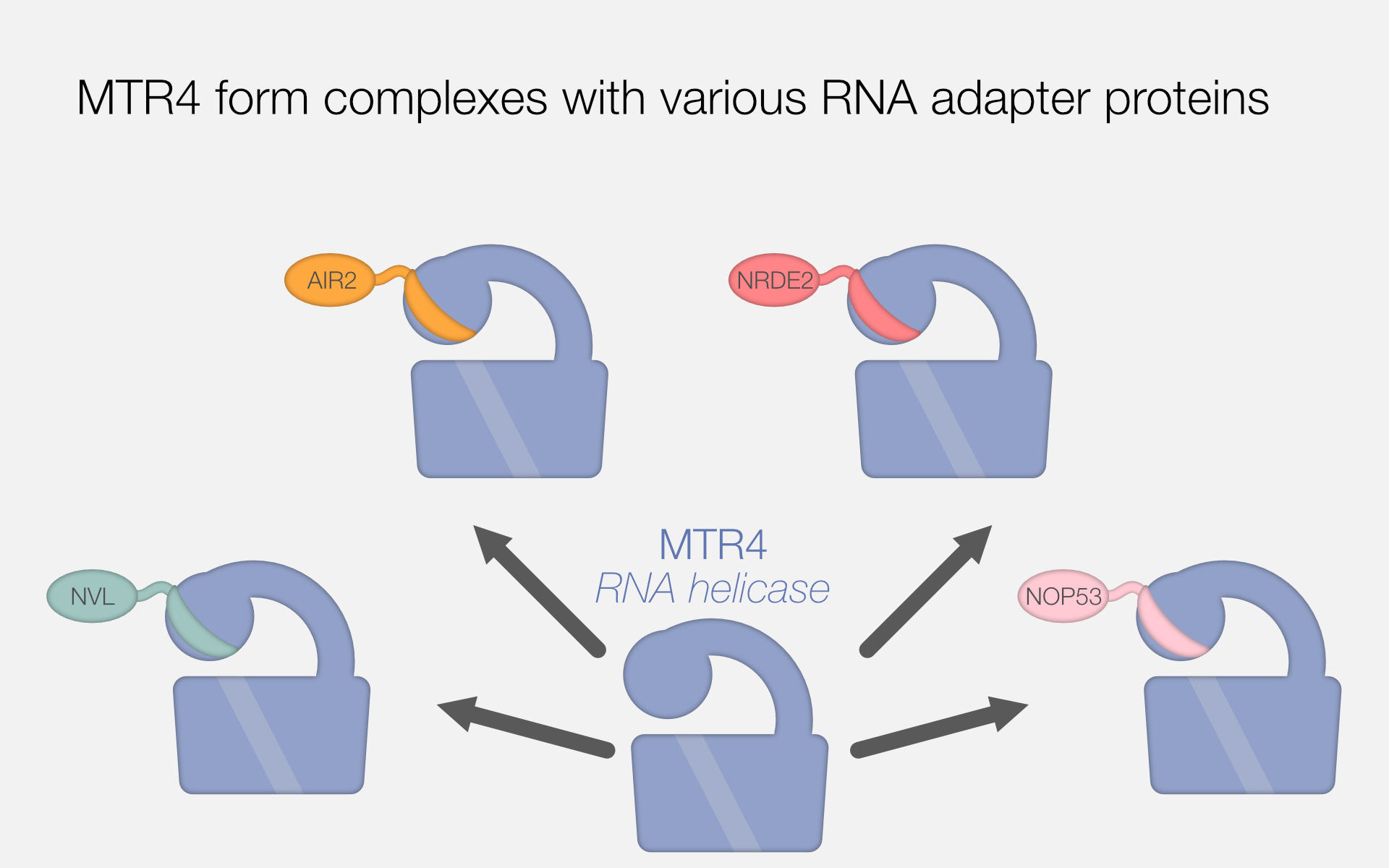
Falk Max Perutz Labs Mtr4 ensures the normal post transcriptional processing of maternal rnas, their nuclear export to the cytoplasm, and the accumulation of properly processed transcripts. oocytes with mtr4 knockout fail to accumulate sufficient and normal transcripts in the cytoplasm and cannot grow to normal sizes. The most striking and unexpected feature of the mtr4 structure is a large, 265 residue domain that arches over the rest of the mtr4 structure and accounts for approximately 25% of the mtr4. The article explores how the human rna helicase mtr4 interacts with different rna binding proteins that function as adaptors for the nuclear exosome complex. it reveals the structural and functional diversity of the adaptors and their binding motifs on mtr4. The saccharomyces cerivisiae mrna transport protein (mtr4) is a rna helicase that interacts with the rna exosome. the exosome core is responsible for processing and degrading rna.
Comments are closed.