Moving Toward Real Time Release Testing

Moving Toward Real Time Release Testing The shift toward real time release testing (rtrt) is changing this long established model. instead of performing all critical checks at the end of the production cycle, rtrt ensures quality throughout the process by relying on continuous monitoring, advanced process control, and real time data. One challenge that continuous processing faces is the difficulties of developing real time alternatives to dissolution that would allow real time release testing (rtrt), promised years ago by fda’s process analytical technology (pat) initiative, to be used with continuous manufacturing.
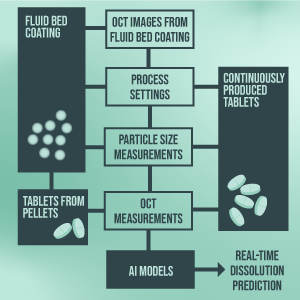
Real Time Release Testing Eccpm This will enable real time release testing (rtrt), which redefines conventional approaches by relying solely on the process data reported by equipment or collected from sensors to predict the product quality. The european commission’s annex 17 revision reflects a transformative vision for pharmaceutical manufacturing — one that prioritizes real time assurance of quality over retrospective testing. Background: the pharmaceutical industry is shifting from end product testing towards real time release testing. this approach is based on the continuous collection of process data and product information, which is finally utilized for the release decision. Real time release testing represents a paradigm shift in quality assurance for biomanufacturing. by enabling product release based on real time process understanding, rtrt improves efficiency, reduces delays, and enhances product quality.

Real Time Release Testing Scientific Guideline European Medicines Background: the pharmaceutical industry is shifting from end product testing towards real time release testing. this approach is based on the continuous collection of process data and product information, which is finally utilized for the release decision. Real time release testing represents a paradigm shift in quality assurance for biomanufacturing. by enabling product release based on real time process understanding, rtrt improves efficiency, reduces delays, and enhances product quality. Release testing ensures pharmaceutical batches meet specifications before distribution. learn qc testing, qp release, skip lot, and real time release methods. Fda’s evolving stance on real time release testing (rtrt): is your qc strategy keeping up? quality control in pharmaceutical manufacturing has long relied on a familiar routine: manufacture the batch, hold it, test the final product, and release it if it passes. This article aims to provide a comprehensive overview of the regulatory expectations surrounding rtrt as outlined in the fda’s process validation. The adoption of real time release testing is transforming pharmaceutical production, driving higher quality and more efficient processes. as the industry continues to evolve, embracing these advancements will be crucial for manufacturers aiming to thrive in a competitive landscape.
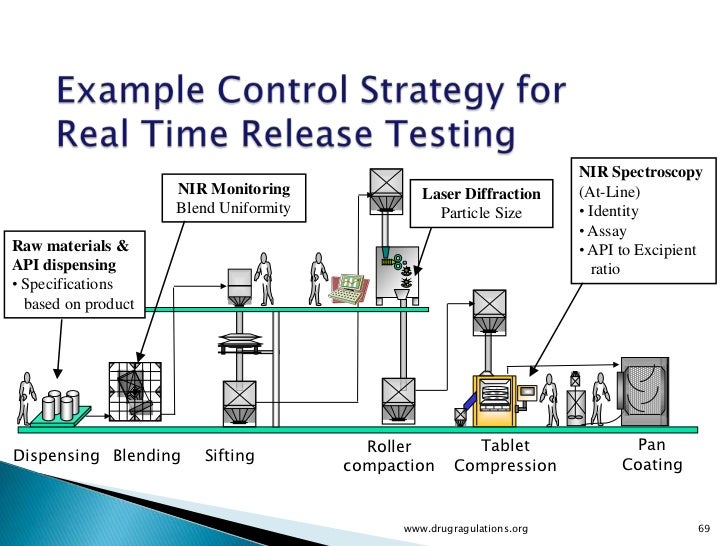
Real Time Release Testing Release testing ensures pharmaceutical batches meet specifications before distribution. learn qc testing, qp release, skip lot, and real time release methods. Fda’s evolving stance on real time release testing (rtrt): is your qc strategy keeping up? quality control in pharmaceutical manufacturing has long relied on a familiar routine: manufacture the batch, hold it, test the final product, and release it if it passes. This article aims to provide a comprehensive overview of the regulatory expectations surrounding rtrt as outlined in the fda’s process validation. The adoption of real time release testing is transforming pharmaceutical production, driving higher quality and more efficient processes. as the industry continues to evolve, embracing these advancements will be crucial for manufacturers aiming to thrive in a competitive landscape.
Comments are closed.