Molecular Orbital Theory 1 Constructive Interference
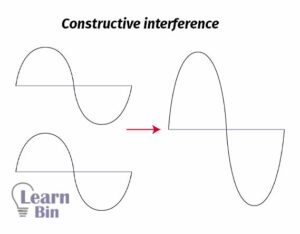
Molecular Orbital Theory Learnbin Bonding molecular orbital: when two atomic orbitals combine so that their wave functions add up, constructive interference is achieved. this creates a bonding molecular orbital, with a high probability of finding electrons between the two nuclei. it helps hold the atoms together in a stable bond. Molecular orbitals are combinations of atomic orbital wave functions. combining waves can lead to constructive interference, in which peaks line up with peaks, or destructive interference, in which peaks line up with troughs (figure 2 2 2).
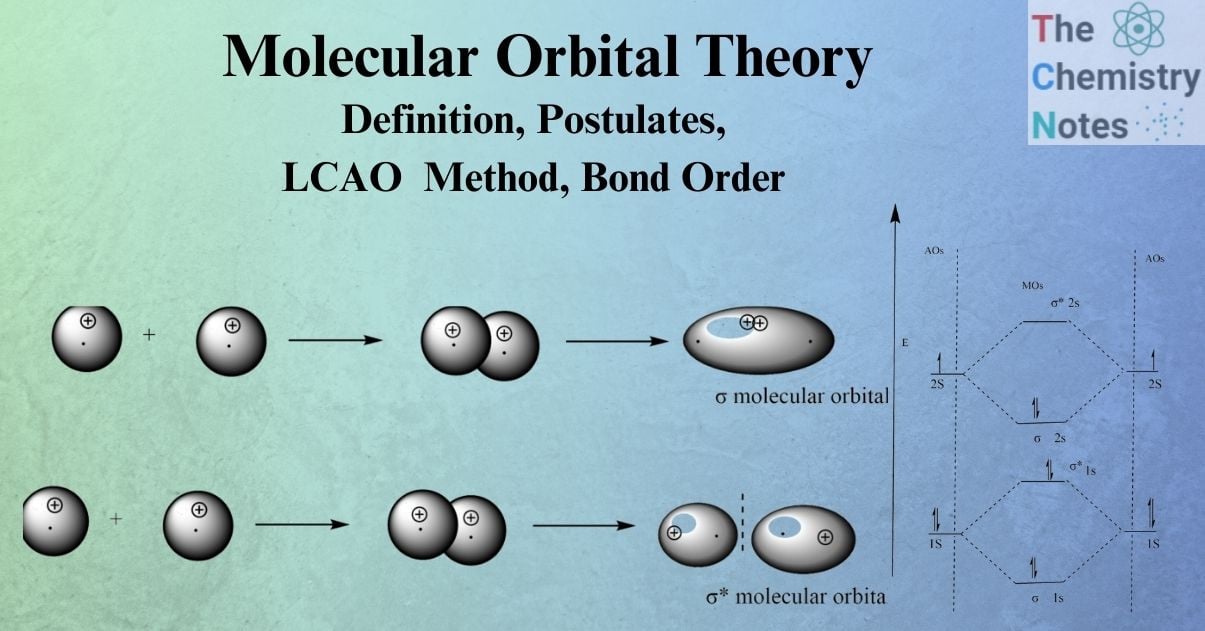
Molecular Orbital Theory Lcao Method Bond Order Constructive interference results in a region of increased amplitude between nuclei, and thus an increased probability density between nuclei (therefore lower energy mo). What is constructive interference? when two waves constructively interfere the resultant wave has an increased amplitude. Molecular orbitals form through the linear combination of atomic orbitals (lcao), where constructive interference creates bonding orbitals (σ, π) (lower energy) and destructive interference forms antibonding orbitals (σ*, π*) (higher energy). 18.1 diatomic molecules atomic orbitals: orbitals that are localized on single atoms. molecular orbitals: orbitals that span two or more atoms. constructing molecular orbitals (mos) by overlapping atomic orbitals (aos) bonds: electron density of mo directed along bond axis.

Precision And Accuracy Molecular orbitals form through the linear combination of atomic orbitals (lcao), where constructive interference creates bonding orbitals (σ, π) (lower energy) and destructive interference forms antibonding orbitals (σ*, π*) (higher energy). 18.1 diatomic molecules atomic orbitals: orbitals that are localized on single atoms. molecular orbitals: orbitals that span two or more atoms. constructing molecular orbitals (mos) by overlapping atomic orbitals (aos) bonds: electron density of mo directed along bond axis. Please, don't forget to hit the like button, subscribe so as to get updated with lots of science materials and share with friends and cl. In molecular orbital theory, we view the bonding of the two h atoms as partial constructive interference between standing wavefunctions of the 1s orbitals. we can also have partial destructive interference. Bonding orbitals are formed when atomic orbitals combine in ways that lead to predominantly constructive interference. the key feature of bonding orbitals is that the molecular orbitals have a lower energy than the corresponding atomic orbitals. Describe how constructive interference is used to explain the formation of bonding and antibonding molecular orbitals in the context of molecular orbital theory.

Solved When Two Atomic Orbitals Combine How Many Molecular Orbitals Please, don't forget to hit the like button, subscribe so as to get updated with lots of science materials and share with friends and cl. In molecular orbital theory, we view the bonding of the two h atoms as partial constructive interference between standing wavefunctions of the 1s orbitals. we can also have partial destructive interference. Bonding orbitals are formed when atomic orbitals combine in ways that lead to predominantly constructive interference. the key feature of bonding orbitals is that the molecular orbitals have a lower energy than the corresponding atomic orbitals. Describe how constructive interference is used to explain the formation of bonding and antibonding molecular orbitals in the context of molecular orbital theory.
Comments are closed.