Molecular And Structural Formula Of Alcohols Alcohol Shorts Methanol Ethanol Class10science
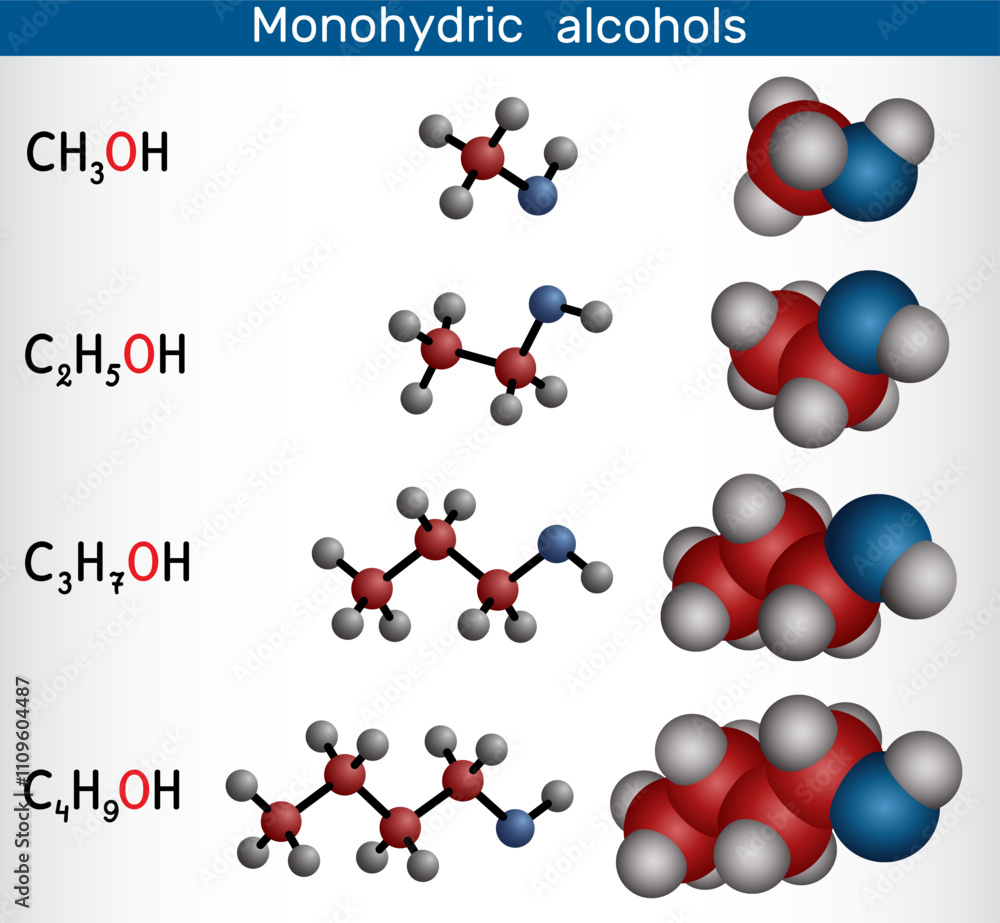
Monohydric Alcohols Methanol Ethanol Propanol Butanol Homologous This chemical's structure consists of a pair of carbon atoms with an alkyl group coupled with an oh functional group; this oh group makes the molecule an alcohol, denoted by the " ol" suffix. When a hydrogen of an alkane.

Structural Chemical Formula Molecular Structure Ethanol Drinking The structure of the molecule of ethanol is ch3−ch2−oh (an ethyl group linked to a hydroxyl group), which indicates that the carbon of a methyl group (−ch3) is attached to the carbon of a methylene group (−ch2−), which is attached to the oxygen of a hydroxyl group (−oh). Subscribed 1.8k 185k views 3 years ago molecular and structural formula of alcohols #alcohol #shorts #methanol #ethanol #class10science more. It also explains the structure of alcohols and lists some basic physical properties of these compounds. alcohol is an organic compound that contains one or more hydroxyl groups. Antoine lavoisier described ethanol as a carbon, hydrogen and oxygen compound, and in 1808 nicolas théodore de saussure determined the chemical formula for ethanol, and in 1858 archibald scott couper released a structural formula for ethanol.

Ethanol Molecule 3d Molecular Structure Ball And Stick Model It also explains the structure of alcohols and lists some basic physical properties of these compounds. alcohol is an organic compound that contains one or more hydroxyl groups. Antoine lavoisier described ethanol as a carbon, hydrogen and oxygen compound, and in 1808 nicolas théodore de saussure determined the chemical formula for ethanol, and in 1858 archibald scott couper released a structural formula for ethanol. Alcohols are covalent molecules; the –oh group in an alcohol molecule is attached to a carbon atom by a covalent bond. we often represent alcohols by the general formula roh, where r is an alkyl group. alcohols are common in nature. Ethanol contains two atoms of carbon, one atom of oxygen, and six atoms of hydrogen. the chemical formula of ethanol is c2h5oh, where oh represents the alcohol functional group. Alcohols are organic compounds attached to a saturated carbon by a hydroxyl group ( oh); that is, a carbon attached to four atoms by simple bonds (without double or triple bonds). the structural formula for alcohol — the vast and versatile family of compounds — is roh. Here, we will study alcohol structure, what is the chemical formula of alcohol, and the application of alcohol in detail. alcohol can be described as having an sp3 hybridized tetrahedral oxygen atom with nonbonding electron pairs occupying two of the four sp3 hybrid orbitals, similar to water.

Ethanol Molecule 3d Molecular Structure Ball And Stick Model Alcohols are covalent molecules; the –oh group in an alcohol molecule is attached to a carbon atom by a covalent bond. we often represent alcohols by the general formula roh, where r is an alkyl group. alcohols are common in nature. Ethanol contains two atoms of carbon, one atom of oxygen, and six atoms of hydrogen. the chemical formula of ethanol is c2h5oh, where oh represents the alcohol functional group. Alcohols are organic compounds attached to a saturated carbon by a hydroxyl group ( oh); that is, a carbon attached to four atoms by simple bonds (without double or triple bonds). the structural formula for alcohol — the vast and versatile family of compounds — is roh. Here, we will study alcohol structure, what is the chemical formula of alcohol, and the application of alcohol in detail. alcohol can be described as having an sp3 hybridized tetrahedral oxygen atom with nonbonding electron pairs occupying two of the four sp3 hybrid orbitals, similar to water.
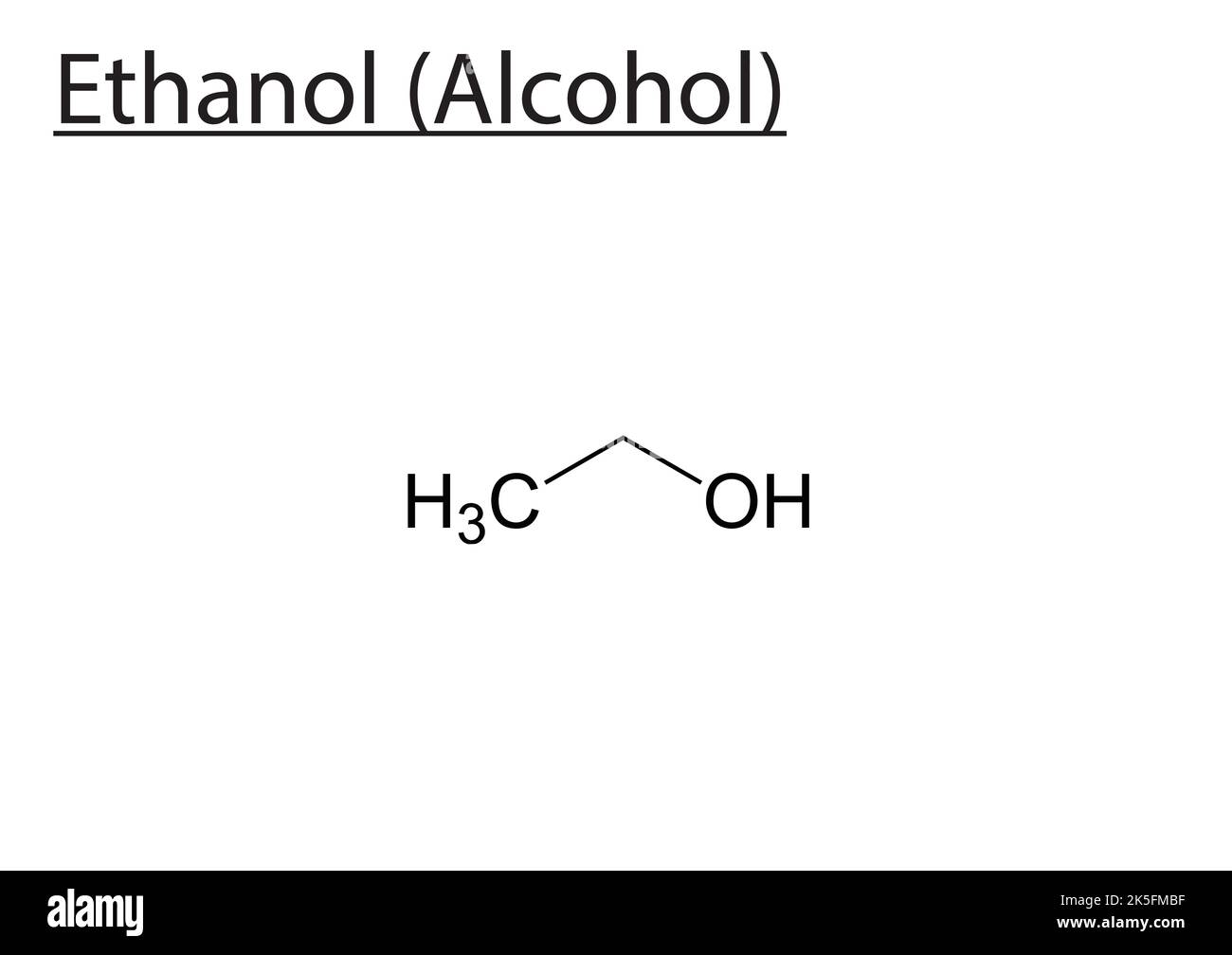
Ethanol Alcohol Molecular Structure Vector Skeletal Formula Stock Alcohols are organic compounds attached to a saturated carbon by a hydroxyl group ( oh); that is, a carbon attached to four atoms by simple bonds (without double or triple bonds). the structural formula for alcohol — the vast and versatile family of compounds — is roh. Here, we will study alcohol structure, what is the chemical formula of alcohol, and the application of alcohol in detail. alcohol can be described as having an sp3 hybridized tetrahedral oxygen atom with nonbonding electron pairs occupying two of the four sp3 hybrid orbitals, similar to water.
Comments are closed.