Mohrs Method

Mohr S Method Introduction Principle Reactions And Calculations Determination of chloride ion concentration by titration (mohr’s method) introduction. The mohr method was first published in 1855 by karl friedrich mohr. because cro 4 2 imparts a yellow color to the solution, which might obscure the end point, only a small amount of k 2 cro 4 is added. as a result, the end point is always later than the equivalence point.
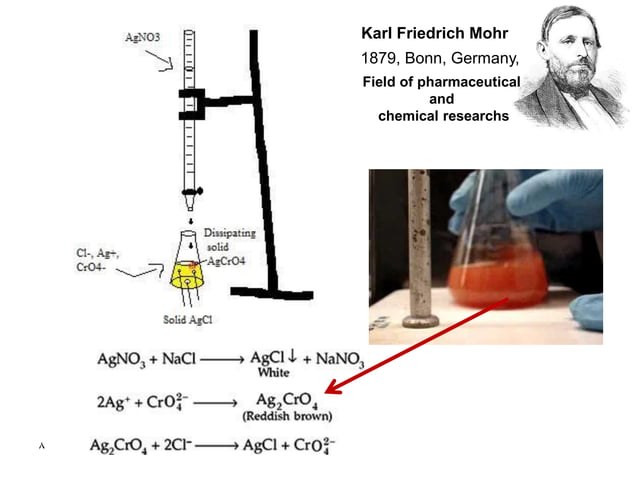
Precepitation Titration Mohrs Method This document describes the mohr method for determining the concentration of chloride ions in an unknown sample through titration with silver nitrate. key steps include: 1) standardizing a silver nitrate solution against sodium chloride to determine its molarity. Mohr method of determination of chlorides by titration with silver nitrate is one of the oldest titration methods still in use it was researched and published by karl friedrich mohr in 1856. It is a quantitative analytic technique that determines the amount of the analyte by measuring volume. mohr’s method uses agno 3 as the titrant (burette) and a small quantity of yellow potassium chromate (k 2 cro 4) as the indicator to determine the concentration of chloride in a given sample. Mohr’s method is a precipitation titration used to determine chloride ion concentrations in a solution. it uses silver nitrate (agno₃) as the titrant, forming a white precipitate of silver chloride (agcl).

Solution Mohrs Method Pptx Studypool It is a quantitative analytic technique that determines the amount of the analyte by measuring volume. mohr’s method uses agno 3 as the titrant (burette) and a small quantity of yellow potassium chromate (k 2 cro 4) as the indicator to determine the concentration of chloride in a given sample. Mohr’s method is a precipitation titration used to determine chloride ion concentrations in a solution. it uses silver nitrate (agno₃) as the titrant, forming a white precipitate of silver chloride (agcl). The method can be used to analyze water samples. it involves titrating aliquots of the sample with a standardized silver nitrate solution until concordant results are obtained. The well known mohr’s method in which alkaline or alkaline earth chlorides react with silver nitrate in the presence of a few drops of potassium chromate solution as indicator is a simple, direct and accurate method for chloride determination. Mohr’s method is based on the formation of an insoluble silver chloride (agcl) or silver bromide (agbr) precipitate when silver nitrate (agno₃) is added to a solution containing chloride (cl⁻) or bromide (br⁻). the titration continues until all the chloride or bromide ions react with the silver ions. Is an indirect method for chloride determination where an excess amount of standard ag is added to the chloride solutioncontainingfe3 as an indicator.

Solution Mohrs Method Pptx Studypool The method can be used to analyze water samples. it involves titrating aliquots of the sample with a standardized silver nitrate solution until concordant results are obtained. The well known mohr’s method in which alkaline or alkaline earth chlorides react with silver nitrate in the presence of a few drops of potassium chromate solution as indicator is a simple, direct and accurate method for chloride determination. Mohr’s method is based on the formation of an insoluble silver chloride (agcl) or silver bromide (agbr) precipitate when silver nitrate (agno₃) is added to a solution containing chloride (cl⁻) or bromide (br⁻). the titration continues until all the chloride or bromide ions react with the silver ions. Is an indirect method for chloride determination where an excess amount of standard ag is added to the chloride solutioncontainingfe3 as an indicator.

Solution Mohrs Method Pptx Studypool Mohr’s method is based on the formation of an insoluble silver chloride (agcl) or silver bromide (agbr) precipitate when silver nitrate (agno₃) is added to a solution containing chloride (cl⁻) or bromide (br⁻). the titration continues until all the chloride or bromide ions react with the silver ions. Is an indirect method for chloride determination where an excess amount of standard ag is added to the chloride solutioncontainingfe3 as an indicator.

Precepitation Titration Mohrs Method Pdf
Comments are closed.