Method Validation Labcompliance Pdf Verification And Validation

Methods Method Verification And Validation Pdf Detection Limit Method validation @ labcompliance.pdf free download as pdf file (.pdf), text file (.txt) or read online for free. validation is the process to confirm that the analytical procedure employed for a specific test is suitable for its intended use. This guideline presents a discussion of elements for consideration during the validation of analytical procedures included as part of registration applications submitted within the ich member regulatory authorities. it provides guidance and recommendations on how to derive and evaluate the various validation tests for each analytical procedure and serves as a collection of terms, and their.

Laboratory Method Verification Pdf This paper is a step by step practical guide for preparing protocols and per forming test methods validation with reference to high performance liquid chromatography (hplc) (use simi lar criteria for all other instrumental test method valida tion) in the quality system compliance industry. To apply an analytical method to a different drug product, appropriate validation or verification studies for compendial procedures with the matrix of the new product should be considered. iii. Requirements to perform a validation or verification study. validations must be performed for all non st. ndard and laboratory developed methods. verifications must be performed for all unmodified standard method. This article gives a review and a strategy for the validation of analytical methods for both in house developed as well as standard methods and a recommendation on the documentation that should be produced during and at the end of method validation.

Method Validation And Verification Pdf Requirements to perform a validation or verification study. validations must be performed for all non st. ndard and laboratory developed methods. verifications must be performed for all unmodified standard method. This article gives a review and a strategy for the validation of analytical methods for both in house developed as well as standard methods and a recommendation on the documentation that should be produced during and at the end of method validation. This paper discusses the critical distinctions between method validation and verification within laboratory practices, emphasizing the need for statistical correlation with established methods. The importance of validation is strengthened, more instructions are issued for the validation of analytical methods (ec 657 2002) as well as the performance criteria (ec 333 2007, ec 401 2006, etc.). “the laboratory shall verify that it can properly perform methods before introducing them by ensuring that it can achieve the required performance. records of the verification shall be retained.”. For the assay method, normally covering from 80% to 120% of the test concentration. for content uniformity, covering minimum of 70% to 130% of the test concentration, based on the nature of the dosage form. for dissolution testing, ±20% over the specified range.

Method Validation Verification Overview Microbiology Lab This paper discusses the critical distinctions between method validation and verification within laboratory practices, emphasizing the need for statistical correlation with established methods. The importance of validation is strengthened, more instructions are issued for the validation of analytical methods (ec 657 2002) as well as the performance criteria (ec 333 2007, ec 401 2006, etc.). “the laboratory shall verify that it can properly perform methods before introducing them by ensuring that it can achieve the required performance. records of the verification shall be retained.”. For the assay method, normally covering from 80% to 120% of the test concentration. for content uniformity, covering minimum of 70% to 130% of the test concentration, based on the nature of the dosage form. for dissolution testing, ±20% over the specified range.
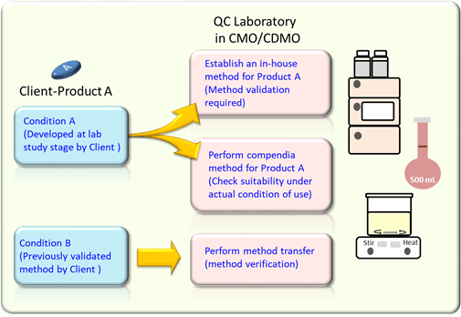
What Is The Difference Between Verification And Validation Of “the laboratory shall verify that it can properly perform methods before introducing them by ensuring that it can achieve the required performance. records of the verification shall be retained.”. For the assay method, normally covering from 80% to 120% of the test concentration. for content uniformity, covering minimum of 70% to 130% of the test concentration, based on the nature of the dosage form. for dissolution testing, ±20% over the specified range.
Comments are closed.