Medical Device Software Development Risks Vs Potentials
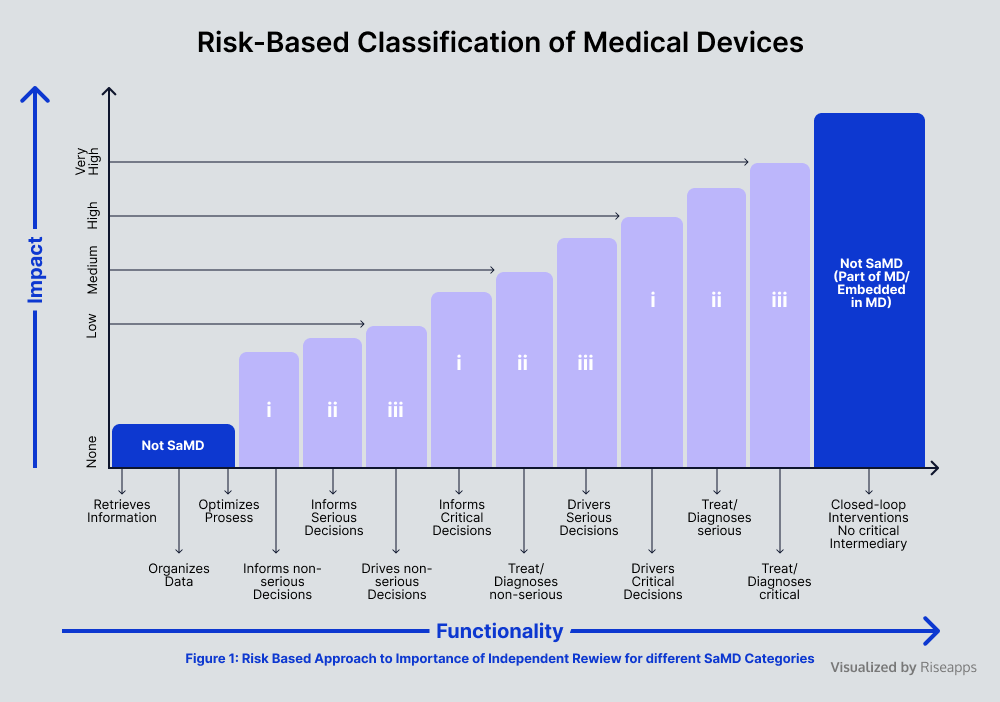
Medical Device Software Development Risks Vs Potentials Cut your medical device software development costs and speed up time to market with the in depth guidebook: top risks, high value features, tips, and more. Jurisdictions may be able to leverage a subset of characterization features and attributes, together with the assessment of medical device software risks and their severity, to describe their approach to applying risk categorization to medical device software.

Medical Device Software Development Risks Vs Potentials This review presents in depth specifications of the stages of the medical device development process and the associated risks, which are not described in organizational or managerial research. Building it is an altogether different task. why? because you are not just coding here, you are actually engineering something that could directly impact a patient’s life. and in this guide, we’ll walk through how medical device software is built, the challenges teams face, and what it really costs to do it right. Effective risk management in samd and medical device software development depends on more than meeting regulatory expectations. it requires full traceability, cross functional alignment, and a unified system that spans the entire product lifecycle. One of the main challenges in managing risks for medical device software is balancing innovation with compliance. developers often face pressure to bring new functionalities to market quickly, which can conflict with the rigorous processes required for risk management.
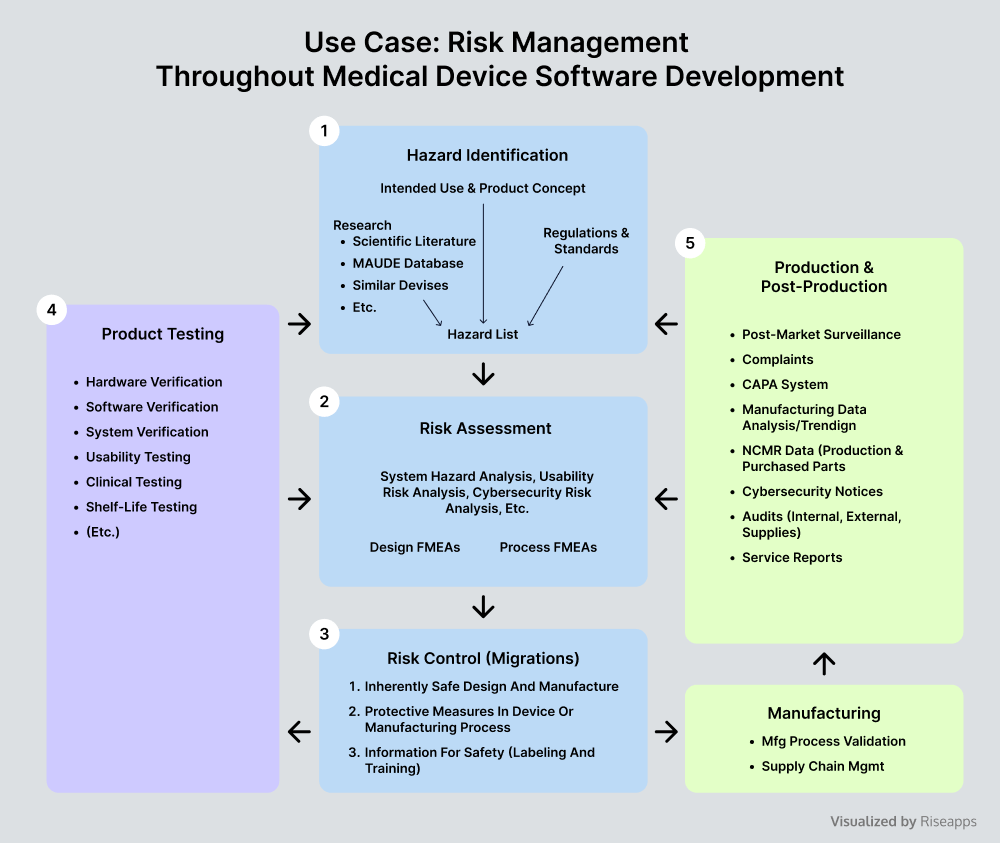
Medical Device Software Development Risks Vs Potentials Effective risk management in samd and medical device software development depends on more than meeting regulatory expectations. it requires full traceability, cross functional alignment, and a unified system that spans the entire product lifecycle. One of the main challenges in managing risks for medical device software is balancing innovation with compliance. developers often face pressure to bring new functionalities to market quickly, which can conflict with the rigorous processes required for risk management. In this post, we’ll cover what medical device software development involves, how it differs from traditional software, and the steps engineers take to build safe, compliant, and reliable systems. This guide explains the essentials of medical software development how to plan, build, and maintain compliant healthcare applications. it focuses on practical challenges, proven approaches, and current trends, without unnecessary jargon or hype. The development of medical device software is one of the most demanding areas in medtech. outdated approaches are no longer sufficient due to the regulatory complexity, interoperability requirements and cybersecurity risks involved. Let’s break down the software development process for medical devices, highlighting crucial aspects that every medical device software developer should consider.
Comments are closed.