Medical Device Reliability Program Pptx

Reliability In Medical Device Industry Pdf Reliability Engineering The document discusses strategies for improving the efficiency and reliability of medical devices through effective risk management and quality control processes. Electric shock, radiation, and environmental protection, care for hygienic factors, heating prevention, mechanical hazard prevention, and material choice safe function: three elements accuracy of measurement, reliability, and warning for or prevention of dangerous outputs.

Exploring Medical Device Reliability And Pdf This program was developed with the intention to elevate medical device stakeholders from the established standard of regulatory compliance to advancing medical device quality and safety through sustained and predictive practices that achieve better patient outcomes. Build a clear risk management plan, align with fda guidelines for medical devices, and track medical device quality assurance and post market surveillance using iso 14971 compliance. Managing medical devices mhra free download as powerpoint presentation (.ppt .pptx), pdf file (.pdf), text file (.txt) or view presentation slides online. the document outlines the systematic approach needed to effectively manage medical devices within a healthcare organization. Can a company avoid recalls for reliability issues by knowing what the reliability will be before product release? an optimal new product development process will be presented that successfully deals with these challenges.

Medical Device Reliability Program Pptx Managing medical devices mhra free download as powerpoint presentation (.ppt .pptx), pdf file (.pdf), text file (.txt) or view presentation slides online. the document outlines the systematic approach needed to effectively manage medical devices within a healthcare organization. Can a company avoid recalls for reliability issues by knowing what the reliability will be before product release? an optimal new product development process will be presented that successfully deals with these challenges. Prognostics in medical devices analyze the data collected from various types of sensors in real time to diagnose performance problems, discern impending faults, and schedule maintenance procedures. Presentation on medical device reliability, classifications, risk management, and regulations. covers device life span and safety. Medical devices reliability and durability 3.1 design for reliability (dfr) is a process that spans the entire product development cycle from concept to release. it increases the probability of delivering a reliable product with the appropriate life cycle cost to the market in a timely manner. It requires continuous monitoring of residual risks and post market information. iso 14971 supports manufacturers in making risk based decisions during design, production, and post market surveillance. overall, it ensures medical devices are safe, effective, and reliable for intended use. download as a pptx, pdf or view online for free.

Medical Device Reliability Program Pptx Prognostics in medical devices analyze the data collected from various types of sensors in real time to diagnose performance problems, discern impending faults, and schedule maintenance procedures. Presentation on medical device reliability, classifications, risk management, and regulations. covers device life span and safety. Medical devices reliability and durability 3.1 design for reliability (dfr) is a process that spans the entire product development cycle from concept to release. it increases the probability of delivering a reliable product with the appropriate life cycle cost to the market in a timely manner. It requires continuous monitoring of residual risks and post market information. iso 14971 supports manufacturers in making risk based decisions during design, production, and post market surveillance. overall, it ensures medical devices are safe, effective, and reliable for intended use. download as a pptx, pdf or view online for free.
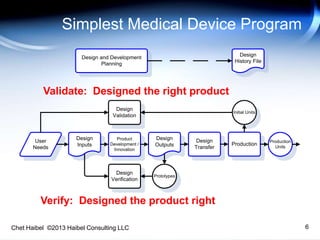
Medical Device Reliability Program Pptx Medical devices reliability and durability 3.1 design for reliability (dfr) is a process that spans the entire product development cycle from concept to release. it increases the probability of delivering a reliable product with the appropriate life cycle cost to the market in a timely manner. It requires continuous monitoring of residual risks and post market information. iso 14971 supports manufacturers in making risk based decisions during design, production, and post market surveillance. overall, it ensures medical devices are safe, effective, and reliable for intended use. download as a pptx, pdf or view online for free.

Medical Device Reliability Program Pptx
Comments are closed.