Medical Device Product Development Lifecycle Support

Medical Device Product Development Life Cycle Pdf Medical Device We support every stage of device development—from concept and design to verification, validation, and usability testing—ensuring your product not only meets global regulatory requirements but performs as intended in the hands of end users. Discover the five phases of the medical device lifecycle, from concept to post market follow up, and learn how to meet regulatory and quality requirements.
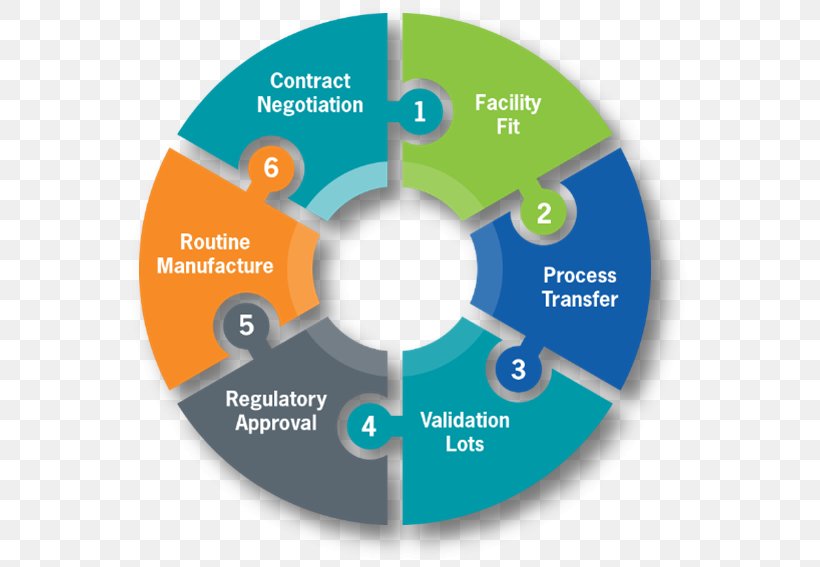
Product Life Cycle Management Medical Device Product Lifecycle This medical device design and development process, guide offers real world insights, strategic advice, and a how to for avoiding costly pitfalls. Learn about chaban medical's complete medical device product lifecycle support and our advanced development and production capabilities. We offer comprehensive lifecycle support services for medical device software. from monitoring and patch management to feature enhancements, recertifications, and adapting to new regulatory requirements, we ensure your device stays safe, effective, and competitive throughout its entire lifecycle. Methodsense aligns regulatory strategy, quality assurance, and documentation support with every phase of your product lifecycle. whether you’re building your first prototype or preparing for post market surveillance, we help you stay on track, on time, and in compliance.

The 4 Phases Of Medical Device Product Development Lifecycle Quasar Med We offer comprehensive lifecycle support services for medical device software. from monitoring and patch management to feature enhancements, recertifications, and adapting to new regulatory requirements, we ensure your device stays safe, effective, and competitive throughout its entire lifecycle. Methodsense aligns regulatory strategy, quality assurance, and documentation support with every phase of your product lifecycle. whether you’re building your first prototype or preparing for post market surveillance, we help you stay on track, on time, and in compliance. Dive into an excellence guide to the medical device product life cycle. explore the iterative process for market ready medical devices through prototyping, testing, and refinement. Overall, the tplc approach allows the fda to review and monitor medical devices throughout their life cycle by taking into account all available information on safety and effectiveness. G&h provides full lifecycle management for medical devices and life science instruments including supply chain continuity, obsolescence management, and ongoing quality assurance. Learn about medical device product development — from concept and design to testing and market release. in every phase, arete biosciences is here to help.
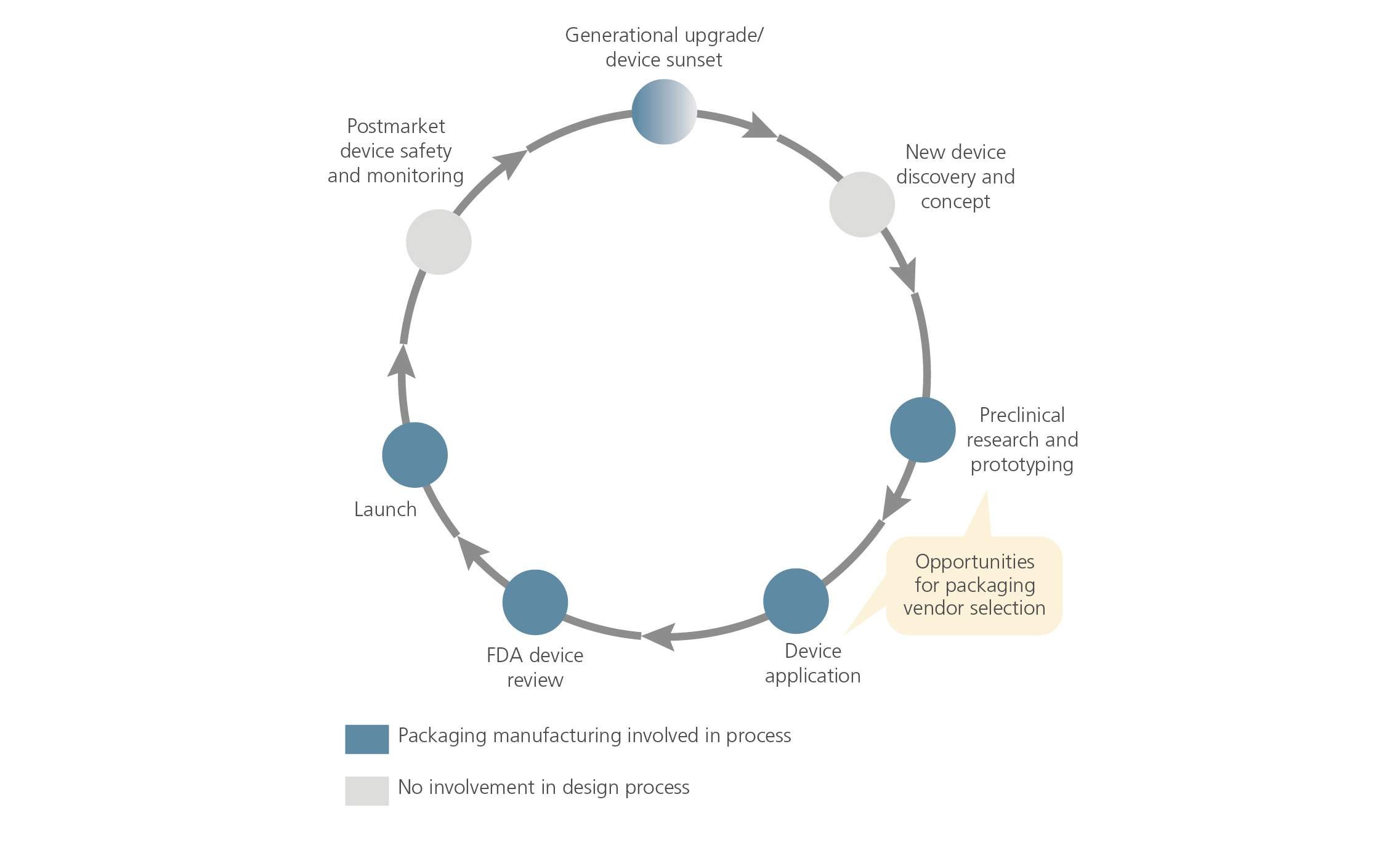
Medical Device Product Lifecycle Management Dive into an excellence guide to the medical device product life cycle. explore the iterative process for market ready medical devices through prototyping, testing, and refinement. Overall, the tplc approach allows the fda to review and monitor medical devices throughout their life cycle by taking into account all available information on safety and effectiveness. G&h provides full lifecycle management for medical devices and life science instruments including supply chain continuity, obsolescence management, and ongoing quality assurance. Learn about medical device product development — from concept and design to testing and market release. in every phase, arete biosciences is here to help.
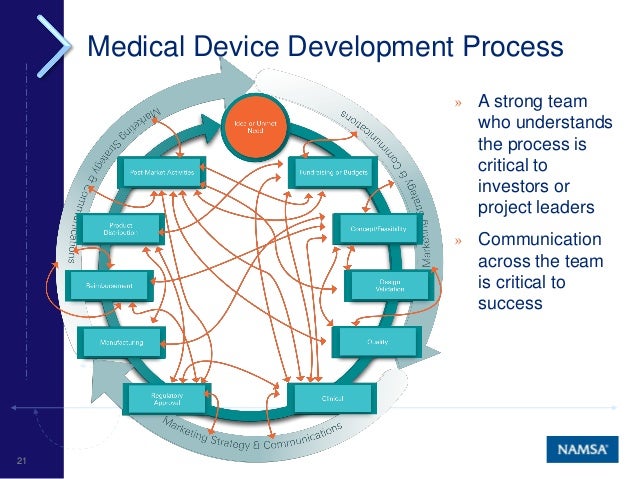
Medical Device Product Lifecycle Management G&h provides full lifecycle management for medical devices and life science instruments including supply chain continuity, obsolescence management, and ongoing quality assurance. Learn about medical device product development — from concept and design to testing and market release. in every phase, arete biosciences is here to help.
Comments are closed.