Medical Device Classification I3cglobal

Medical Device Classification Pdf Medical Device Clinical Medicine Eu mdr medical device classification this page provides a concise overview of the medical device classification system under the mdr 2017 745 regulation. it offers examples of devices within each class and indicates the corresponding type of certification required. Before affixing medical device ce marking, the device must follow the definition and be classified correctly following the classification rules laid down in annexes viii of the eu mdr 2017 745. medical devices are divided into four risk classes: i (im, is), iia, iib and iii.
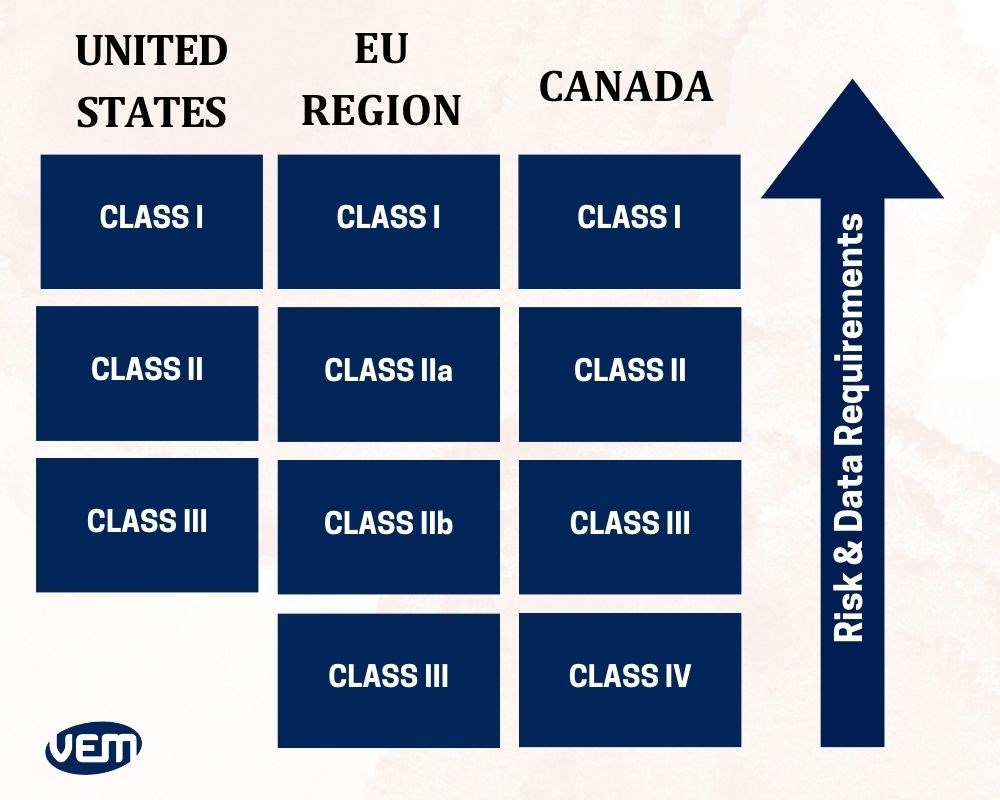
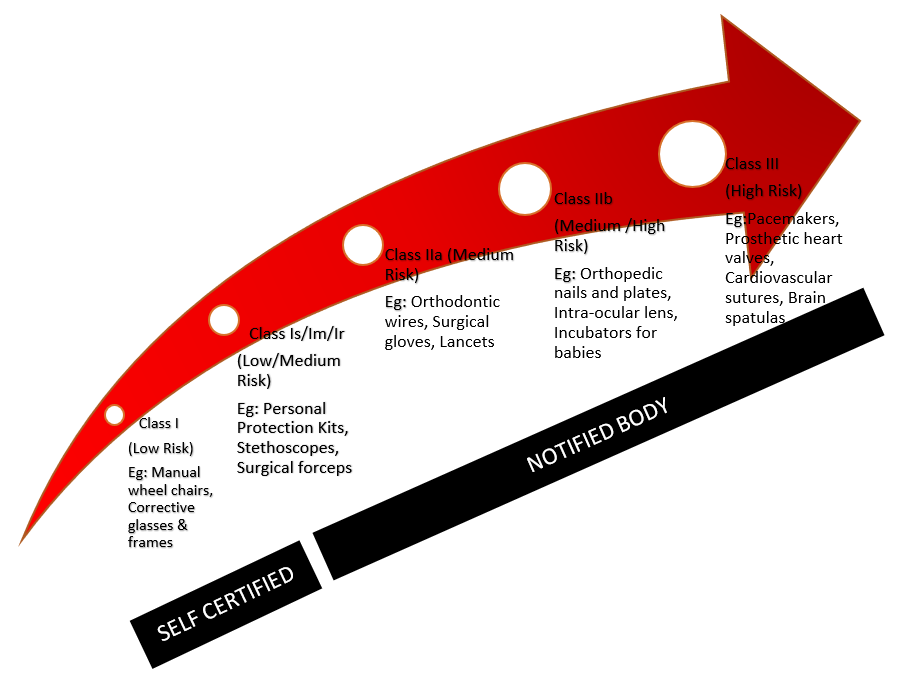
Guide To Medical Device Classification There are basically four classes, ranging from low risk to high risk. a medical device (md) may be classified as class 1** (including is & im), iia**, iib** and iii**, with class iii covering the highest risk products. the higher the classification the greater the level of assessment required. Viii of regulation (eu) 2017 745 on medical devices (mdr). they correspond, to a large extent, to the classification rules established by the international medical device regulators. This database includes: a list of all medical devices with their associated classifications, product codes, fda premarket review organizations, and other regulatory information. learn more. Medical device classification system is used to assess potential risks associated with the medical device and to decide the regulatory pathway to ensure quality standards are met.

Medical Device Classification Guide This database includes: a list of all medical devices with their associated classifications, product codes, fda premarket review organizations, and other regulatory information. learn more. Medical device classification system is used to assess potential risks associated with the medical device and to decide the regulatory pathway to ensure quality standards are met. In this guide, we present the different classes for medical devices, explain how medical devices must be classified, delve into the classification rules, and explore the implications of each classification. This document applies to all products that fall within the definition of the term ‘medical device’, other than ivd medical devices, for which a separate classification document is available on the ghtf website. This meddev contains guidance for the application of the classification rules for medical devices as set out in annex ix of directive 93 42 eec1, as amended. it is for the national competent authorities and national courts to take legally binding decisions on a case by case basis. The normative requirements for active medical devices, which include medical electrical equipment, are documented under the iec 60601 standards family and include the requirements for accessing many global markets under medical device directive (mdd) 93 42 eec.

Uk Medical Device Classification In this guide, we present the different classes for medical devices, explain how medical devices must be classified, delve into the classification rules, and explore the implications of each classification. This document applies to all products that fall within the definition of the term ‘medical device’, other than ivd medical devices, for which a separate classification document is available on the ghtf website. This meddev contains guidance for the application of the classification rules for medical devices as set out in annex ix of directive 93 42 eec1, as amended. it is for the national competent authorities and national courts to take legally binding decisions on a case by case basis. The normative requirements for active medical devices, which include medical electrical equipment, are documented under the iec 60601 standards family and include the requirements for accessing many global markets under medical device directive (mdd) 93 42 eec.
Medical Device Classification I3cglobal This meddev contains guidance for the application of the classification rules for medical devices as set out in annex ix of directive 93 42 eec1, as amended. it is for the national competent authorities and national courts to take legally binding decisions on a case by case basis. The normative requirements for active medical devices, which include medical electrical equipment, are documented under the iec 60601 standards family and include the requirements for accessing many global markets under medical device directive (mdd) 93 42 eec.
Comments are closed.