Mass Changes With Gases Study
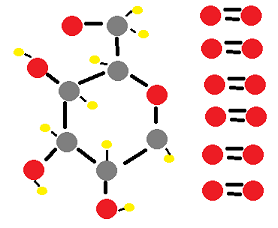
Mass Changes With Gases Study Use our notes to learn how mass changes when a reactant or product is a gas during a reaction. then use our exam style questions to test yourself. learn more. In this study, a quasi static closed tank experiment is conducted to explore the static gas to liquid and liquid to gas mass transfer characteristics. during the pressurization process (gas injection), the liquid is unsaturated and the gas is in a supersaturated state.
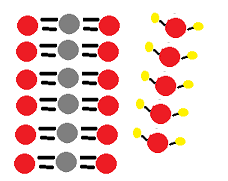
Mass Changes With Gases Study We have previously measured quantities of reactants and products using masses for solids and volumes in conjunction with the molarity for solutions; now we can also use gas volumes to indicate quantities. The goal of this chapter is to review the laws of chemistry and physics that govern the behavior of gases. the material in this chapter will help you develop a mental picture, or model, of the behavior of gases so that you will be able to visualize what is actually happening inside a vacuum system. 2.2 states of matter. In this lesson, we will discover why some reactions seem to disobey the law that matter cannot be created or destroyed because the starting and ending mass appear to be different. In this chapter, we explore the relationships among pressure, temperature, volume, and the amount of gases. you will learn how to use these relationships to describe the physical behavior of a sample of both a pure gaseous substance and mixtures of gases.
/143058836-56a12f0a5f9b58b7d0bcdab4.jpg)
Chemistry Study Guide For Gases In this lesson, we will discover why some reactions seem to disobey the law that matter cannot be created or destroyed because the starting and ending mass appear to be different. In this chapter, we explore the relationships among pressure, temperature, volume, and the amount of gases. you will learn how to use these relationships to describe the physical behavior of a sample of both a pure gaseous substance and mixtures of gases. This course is concerned solely with the properties and behaviour of gases. as we shall see, the fact that interactions between gas phase particles are only very weak allows us to use relatively simple models to gain virtually a complete understanding of the gas phase. Comprehensive general chemistry study guide covering gas laws, kinetic molecular theory, real gases, and calculations for exams and homework. This topic focuses on how the mass of reactants and products can change, especially when gases are involved. we'll break it down, so you feel confident and prepared for your exam. Perform stoichiometric calculations for gas phase reactions. relate the mole fraction and partial pressure and partial volume of a particular gas in a mixture to its contribution to the total pressure and total volume.
Mass Changes In Chemical Reactions This course is concerned solely with the properties and behaviour of gases. as we shall see, the fact that interactions between gas phase particles are only very weak allows us to use relatively simple models to gain virtually a complete understanding of the gas phase. Comprehensive general chemistry study guide covering gas laws, kinetic molecular theory, real gases, and calculations for exams and homework. This topic focuses on how the mass of reactants and products can change, especially when gases are involved. we'll break it down, so you feel confident and prepared for your exam. Perform stoichiometric calculations for gas phase reactions. relate the mole fraction and partial pressure and partial volume of a particular gas in a mixture to its contribution to the total pressure and total volume.

Changes In The Mass Intensity Of Major Gases During The Pyrolysis Of This topic focuses on how the mass of reactants and products can change, especially when gases are involved. we'll break it down, so you feel confident and prepared for your exam. Perform stoichiometric calculations for gas phase reactions. relate the mole fraction and partial pressure and partial volume of a particular gas in a mixture to its contribution to the total pressure and total volume.

Study Of Colored Gases In The Laboratory Stock Illustration
Comments are closed.