Lysine At Ph 7

Lysine At Ph 7 Two pathways are responsible for the de novo biosynthesis of l lysine, namely the (a) diaminopimelate pathway and (b) α‑aminoadipate pathway. two pathways have been identified in nature for the synthesis of lysine. At ph 7 lysine has a net charge of very close to 1. the carboxylic acid group is fully deprotonated ( 1 charge). the alpha amino group is about 99% protonated ( 1 charge). the side chain amino group is fully protonated ( 1 charge).

Lysine At Ph 7 Depending on the degree of protonation of these functional groups (nh2 ↔ nh3 , cooh ↔ coo − ), the net charge of llysine changes greatly (positive, neutral or negative) as a function of ph in. At physiological ph, lysine has a net positive charge. thus, we have to increase the ph of the solution to remove positive charge in order to reach the isoelectric point. The carboxyl groups of the side chains have pk values of 3.9 (asp) and 4.3 (glu), so these side chains are ionized at ph 7. the side chain of lysine is a hydrophobic chain of four methylene groups (the β, γ, δ, and ε carbon atoms) capped by an ε amino group. For these amino acids, the protonated forms predominate at physiological ph (about 7). two amino acids have acidic side chains at neutral ph. these are aspartic acid or aspartate (asp) and glutamic acid or glutamate (glu).
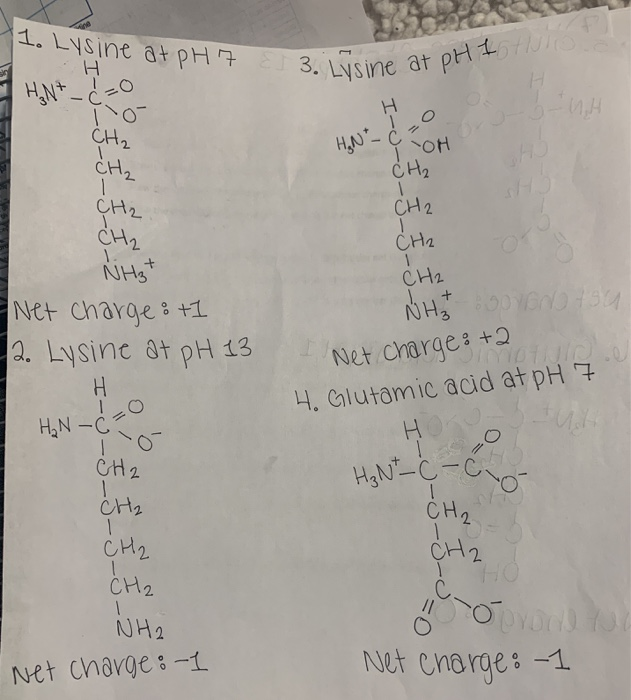
Solved 1 Lysine At Ph7 E 3 Lysine At Ph Itno H N C Ch2 Chegg The carboxyl groups of the side chains have pk values of 3.9 (asp) and 4.3 (glu), so these side chains are ionized at ph 7. the side chain of lysine is a hydrophobic chain of four methylene groups (the β, γ, δ, and ε carbon atoms) capped by an ε amino group. For these amino acids, the protonated forms predominate at physiological ph (about 7). two amino acids have acidic side chains at neutral ph. these are aspartic acid or aspartate (asp) and glutamic acid or glutamate (glu). The optimal ph for the stability and function of lysine in biological systems is around ph 7. lysine is most stable and functions best at this neutral ph level. At a physiological ph of 7.2, lysine has a net positive charge due to its protonated side chain, while glutamate has a net negative charge as its side chain is deprotonated. The amino acids are referred to as hydrophilic amino acids and include arginine, lysine, aspartic acid, and glutamic acid. at ph 7 the side chains of these amino acids carry charges—positive for arginine and lysine, negative. At physiological ph (around 7.4), the ε amino group is predominantly protonated, giving lysine a net positive charge. however, as the ph increases, the ε amino group becomes deprotonated, and the net charge of lysine shifts towards neutral or negative.

Lysine Structure At Different Ph Frontiers Sodium Ions Affect The optimal ph for the stability and function of lysine in biological systems is around ph 7. lysine is most stable and functions best at this neutral ph level. At a physiological ph of 7.2, lysine has a net positive charge due to its protonated side chain, while glutamate has a net negative charge as its side chain is deprotonated. The amino acids are referred to as hydrophilic amino acids and include arginine, lysine, aspartic acid, and glutamic acid. at ph 7 the side chains of these amino acids carry charges—positive for arginine and lysine, negative. At physiological ph (around 7.4), the ε amino group is predominantly protonated, giving lysine a net positive charge. however, as the ph increases, the ε amino group becomes deprotonated, and the net charge of lysine shifts towards neutral or negative.

Lysine Structure At Different Ph Frontiers Sodium Ions Affect The amino acids are referred to as hydrophilic amino acids and include arginine, lysine, aspartic acid, and glutamic acid. at ph 7 the side chains of these amino acids carry charges—positive for arginine and lysine, negative. At physiological ph (around 7.4), the ε amino group is predominantly protonated, giving lysine a net positive charge. however, as the ph increases, the ε amino group becomes deprotonated, and the net charge of lysine shifts towards neutral or negative.
Comments are closed.