Limiting Reactant Problem Set Pdf Science Mathematics

Limiting Reactant Key Pdf This document provides 5 examples of stoichiometry calculations involving limiting reactants. the examples include reactions between lithium and oxygen, aluminum and copper nitrate, ammonia and copper oxide, zinc and silver nitrate solution, and naphthalene oxidation. 2c2h2 5o2 → 2h2o 4co2 (a) identify the limiting reactant. (b) how many moles of carbon dioxide gas (co2) will form? 3. a student places 2.36 moles of acetic acid (ch3co2h) and 3.89 moles of sodium hydroxide (naoh) in a beaker of water. they react to form sodium acetate (nach3co2) and water. ch3co2h naoh → nach3co2 h2o.

Limiting Reactant Stoichiometry A Chemistry Worksheet Made By Why practice limiting reactant problems regularly? limiting reactant problems are more than just academic exercises; they teach critical thinking and problem solving skills that are applicable across scientific disciplines. understanding how to identify the limiting reactant not only improves your grasp of chemical reactions but also prepares you for real world applications such as chemical. Downloadable pdf guides offer comprehensive problem sets with detailed solutions, focusing on limiting reagent calculations. these resources include step by step explanations, examples, and exercises to enhance understanding and problem solving skills in stoichiometry and chemical reactions. 10 targeted problems: the worksheet features ten stoichiometry problems that cover key concepts, including limiting reactants and percent yield. full answer key: a detailed, step by step answer key is included, allowing you to check your work and understand the logic behind each solution. O2 would be the limiting reactant because it only makes 1.125 moles of h2o before it is used up.

Post Lab 8 Limiting Reactant Pdf 10 targeted problems: the worksheet features ten stoichiometry problems that cover key concepts, including limiting reactants and percent yield. full answer key: a detailed, step by step answer key is included, allowing you to check your work and understand the logic behind each solution. O2 would be the limiting reactant because it only makes 1.125 moles of h2o before it is used up. Try 3 problem set limiting reactants pedersenscience 1. when 114.0 g of iron. 29. .7 g of chlorine gas reacts, iron(iii) chloride . fo. med. a. how many grams of iron(iii) chloride are formed? d. . ow. uch excess reagent is left over at the end of the experiment? 2. in a combustion react. on. Silver nitrate is the limiting reagent. let’s stop for a moment and look at what that means in this problem. silver nitrate reacts with calcium chloride in a ratio of 2 : 1. we would expect 0.104 mol of agno3 to react completely with 0.0520 mol of cacl2, since that’s also a 2 : 1 ratio. Practice problems: limiting reagents 1. take the reaction: nh3 o2 no h2o. in an experiment, 3.25 g of nh3 are allowed to react with 3.50 g of o2. a. which reactant is the limiting reagent? b. how many grams of no are formed?. Use coefficient ratios and availability ratios to think through what is limiting and excess and to complete the math on your own. but if needed, use the simulation to complete or to check answers for the tables.
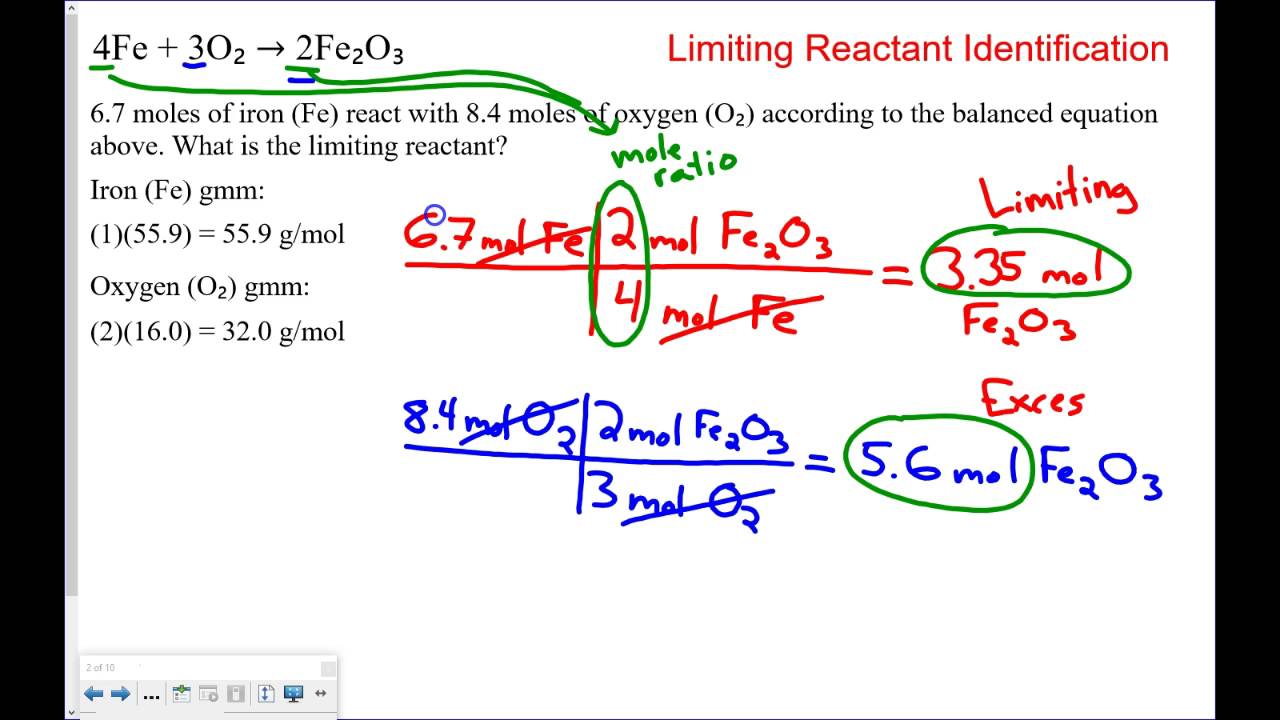
Excess Reactant Limiting Reactants Mr Gary S Science Class Kam Try 3 problem set limiting reactants pedersenscience 1. when 114.0 g of iron. 29. .7 g of chlorine gas reacts, iron(iii) chloride . fo. med. a. how many grams of iron(iii) chloride are formed? d. . ow. uch excess reagent is left over at the end of the experiment? 2. in a combustion react. on. Silver nitrate is the limiting reagent. let’s stop for a moment and look at what that means in this problem. silver nitrate reacts with calcium chloride in a ratio of 2 : 1. we would expect 0.104 mol of agno3 to react completely with 0.0520 mol of cacl2, since that’s also a 2 : 1 ratio. Practice problems: limiting reagents 1. take the reaction: nh3 o2 no h2o. in an experiment, 3.25 g of nh3 are allowed to react with 3.50 g of o2. a. which reactant is the limiting reagent? b. how many grams of no are formed?. Use coefficient ratios and availability ratios to think through what is limiting and excess and to complete the math on your own. but if needed, use the simulation to complete or to check answers for the tables.

Solved Solving Another Limiting Reactant Problem Using A Chegg Practice problems: limiting reagents 1. take the reaction: nh3 o2 no h2o. in an experiment, 3.25 g of nh3 are allowed to react with 3.50 g of o2. a. which reactant is the limiting reagent? b. how many grams of no are formed?. Use coefficient ratios and availability ratios to think through what is limiting and excess and to complete the math on your own. but if needed, use the simulation to complete or to check answers for the tables.
Comments are closed.