Lifecycle Approach To Api Process Validation

Lifecycle Api Process Validation Video Regis Technologies If you are committed to transforming your approach to process validation, i invite you to read on. under the life cycle approach, process validation is no longer a finite activity conducted prior to commercialization. instead, it is an ongoing exercise where a process is always in one of three stages [table 1]. Ispe practical implementation of the lifecycle approach to process validation. the ispe good practice guide provides detailed guidance for pharmaceutical companies on the lifecycle approach to process validation, helping them meet global regulatory expectations.

Api Lifecycle Apicontext According to the modern approach, the process validation exercise is divided into 3 stages. figure 1 illustrates the stages and sub stages of process validation. In the following sections, we describe general considerations for process validation, the recommended stages of process validation, and specific activities for each stage in the product. Learn how to design an effective api process validation strategy. understand fda ich stages, phase appropriate requirements, and common pitfalls to avoid. Fda’s 2011 process validation guidance emphasizes a lifecycle approach (development through commercialization) to achieve “a high degree of assurance” that the api will meet its quality.

Validation Life Cycle Presentationeze Learn how to design an effective api process validation strategy. understand fda ich stages, phase appropriate requirements, and common pitfalls to avoid. Fda’s 2011 process validation guidance emphasizes a lifecycle approach (development through commercialization) to achieve “a high degree of assurance” that the api will meet its quality. The document outlines a lifecycle approach to process validation in pharmaceutical manufacturing, detailing stages as defined by fda guidance: process design, process qualification, and continued process verification. Process validation involves a series of activities taking place over the lifecycle of the product and process: process design (stage 1), process qualification (stage 2), and continued process verification (stage 3). The list below summarizes the information needed to make the transition from stage 1 (process design) to stage 2 (performance qualification) in the process validation lifecycle. Explore the fda's lifecycle approach to process validation in pharmaceuticals. learn about process design, qualification, and verification.

The Lifecycle Approach To Process Validation Overview Orioled Hub The document outlines a lifecycle approach to process validation in pharmaceutical manufacturing, detailing stages as defined by fda guidance: process design, process qualification, and continued process verification. Process validation involves a series of activities taking place over the lifecycle of the product and process: process design (stage 1), process qualification (stage 2), and continued process verification (stage 3). The list below summarizes the information needed to make the transition from stage 1 (process design) to stage 2 (performance qualification) in the process validation lifecycle. Explore the fda's lifecycle approach to process validation in pharmaceuticals. learn about process design, qualification, and verification.
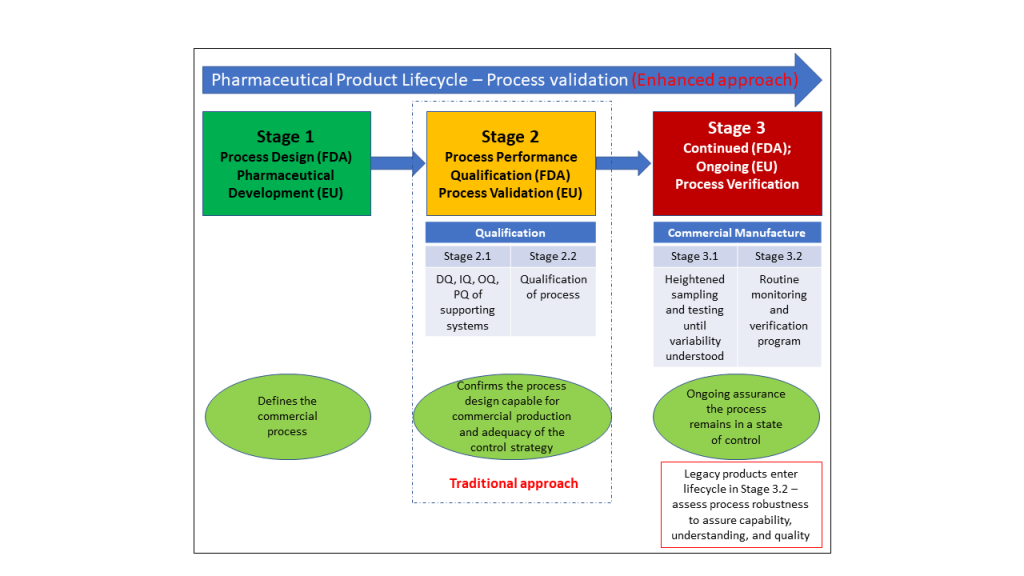
The Lifecycle Approach To Process Validation Overview Orioled Hub The list below summarizes the information needed to make the transition from stage 1 (process design) to stage 2 (performance qualification) in the process validation lifecycle. Explore the fda's lifecycle approach to process validation in pharmaceuticals. learn about process design, qualification, and verification.

The Lifecycle Approach To Process Validation Overview Orioled Hub
Comments are closed.