Lesson 8 Covalent Bond

Lesson 2 Covalent Bond Pdf Chemical Bond Chemical Compounds Covalent bonds form when atoms share one or more pairs of electrons. sharing one pair, two pairs, and three pairs of electrons forms single, double, and triple covalent bonds, respectively. orbitals overlap directly in sigma bonds. There are three main types of bonding: ionic, metallic, and covalent. covalent bonding is the focus of this chapter and forms molecules by electron sharing between nonmetals.

Lesson 16 Pdf Chemical Bond Covalent Bond 8th grade science lesson plan on chemical bonds: ionic and covalent bonding. includes modeling activities, materials, and student objectives. Covalent bonds involve shared electron pairs between atoms. each atom contributes one electron to each shared pair, and effectively gains an additional electron from the shared pair. atoms share the same number of pairs needed to fill their valence shell, usually with eight. Chemistry: matter and change chapter 8: covalent bonding in this chapter: weblinks standardized test practice. The atoms in a molecular compound are held together by covalent bonds. molecular compounds can be represented by molecular formulas, which tell how many of each type of atom are in the compound.

Covalent Bonding And Molecular Formulas Pdf Chemical Bond Molecules Chemistry: matter and change chapter 8: covalent bonding in this chapter: weblinks standardized test practice. The atoms in a molecular compound are held together by covalent bonds. molecular compounds can be represented by molecular formulas, which tell how many of each type of atom are in the compound. On this page, you'll learn how atoms form covalent bonds through electron sharing, understand the octet rule, and distinguish between polar and nonpolar bonds. the graph at the right shows the potential energy of two atoms as a function of the distance that separates the two atoms. In ionic compounds, electrons are transferred between atoms of different elements to form ions. but this is not the only way that compounds can be formed. atoms can also make chemical bonds by sharing electrons between each other. such bonds are called covalent bonds. Lewis dot structures of covalent compounds • in covalent compounds atoms share electrons. we can use lewis structures to help visualize the molecules. Covalent bonds form when two nonmetal atoms share one or more pairs of valence electrons to achieve a noble gas configuration. this sharing of electrons results in the formation of molecules with lower potential energy than the individual atoms.
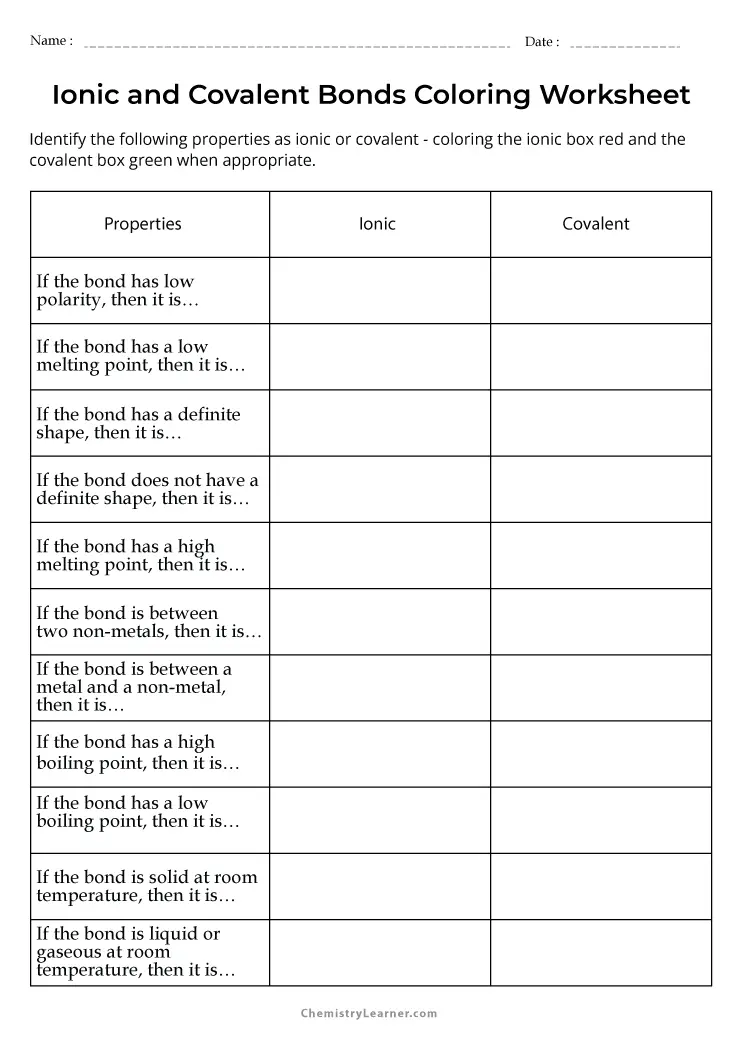
Free Printable Ionic And Covalent Bonds Worksheets Worksheets Library On this page, you'll learn how atoms form covalent bonds through electron sharing, understand the octet rule, and distinguish between polar and nonpolar bonds. the graph at the right shows the potential energy of two atoms as a function of the distance that separates the two atoms. In ionic compounds, electrons are transferred between atoms of different elements to form ions. but this is not the only way that compounds can be formed. atoms can also make chemical bonds by sharing electrons between each other. such bonds are called covalent bonds. Lewis dot structures of covalent compounds • in covalent compounds atoms share electrons. we can use lewis structures to help visualize the molecules. Covalent bonds form when two nonmetal atoms share one or more pairs of valence electrons to achieve a noble gas configuration. this sharing of electrons results in the formation of molecules with lower potential energy than the individual atoms.

Lesson 8 3 Covalent Bonds Lewis dot structures of covalent compounds • in covalent compounds atoms share electrons. we can use lewis structures to help visualize the molecules. Covalent bonds form when two nonmetal atoms share one or more pairs of valence electrons to achieve a noble gas configuration. this sharing of electrons results in the formation of molecules with lower potential energy than the individual atoms.
Comments are closed.