Lesson 3 History Of Periodic Table Pdf Chemical Elements

History Of The Periodic Table Of Elements Pdf Chemical Elements Lesson 3 history of periodic table free download as pdf file (.pdf), text file (.txt) or read online for free. The table’s form has evolved over the decades. starting with mendeleev’s first published work on the periodic table in 1869, there were question marks noting gaps in knowledge, and copious disagreements on what should be placed where and why.

Dll History Of The Periodic Table Pdf Periodic Table Chemical Moseley noticed that when all the elements were arranged in order of mass a few were not in the right family with respect to properties moseley used a procedure called x ray diffraction to determine the atomic number of the elements. During the next 200 years, a vast body of knowledge concerning the properties of elements and their compounds was acquired by chemists (view a 1790 article on the elements). by 1869, a total of 63 elements had been discovered. Evolution of the periodic table ntury, if the periodic table were constructed, it would have looked like figure 1. towards the beginning of the 19th century, many scientists th ught of the con cept of periodicity of periodic properties of elements (figure 2). the first one to examine this was dobereine. In 1869, dmitri mendeleev created the first periodic table and ordered the elements by their atomic masses. he noticed that elements in the same groups had similar chemical and physical properties.

Chapter 1 Lesson 3 Periodic Table Of Elements Pdf Evolution of the periodic table ntury, if the periodic table were constructed, it would have looked like figure 1. towards the beginning of the 19th century, many scientists th ught of the con cept of periodicity of periodic properties of elements (figure 2). the first one to examine this was dobereine. In 1869, dmitri mendeleev created the first periodic table and ordered the elements by their atomic masses. he noticed that elements in the same groups had similar chemical and physical properties. Elements have a unique set of spectral lines that allows us to identify them this is how we know the sun contains h and he, even though we’ve never been there. Draw the complete electron configuration of each of the following elements. what ions will they form? when ions, how many electrons does each have? how many protons? predict the relative diameters of the members of this isoelectronic series. 2. atomic radius. The physical and chemical properties of elements are the periodic function of their atomic weights, i.e., when the elements are arranged in order of their increasing atomic weights, elements with similar properties are repeated after certain regular intervals. After completion of this activity, students should have a clear understanding of how the periodic table is used and the way it organizes the elements by not only atomic number but by the characteristics of each element as a whole.
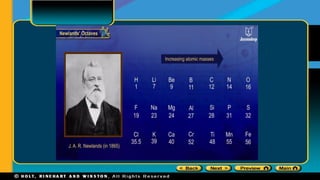
5 Lesson 1 History Of The Periodic Table Pptx Elements have a unique set of spectral lines that allows us to identify them this is how we know the sun contains h and he, even though we’ve never been there. Draw the complete electron configuration of each of the following elements. what ions will they form? when ions, how many electrons does each have? how many protons? predict the relative diameters of the members of this isoelectronic series. 2. atomic radius. The physical and chemical properties of elements are the periodic function of their atomic weights, i.e., when the elements are arranged in order of their increasing atomic weights, elements with similar properties are repeated after certain regular intervals. After completion of this activity, students should have a clear understanding of how the periodic table is used and the way it organizes the elements by not only atomic number but by the characteristics of each element as a whole.
Comments are closed.