Lab Incident Reporting Software Korea 2025 Amplelogic

Korea Lab 2025 Krüss Scientific Explore amplelogic's lab incident reporting software korea for 2025. streamline incident management and ensure laboratory safety. Electronic quality management system built on low code apaas platform, providing 11 integrated modules for pharmaceutical and life sciences quality management with ai powered automation and 10x faster deployment.

Lab Incident Reporting Software Spain 2024 Amplelogic Read verified amplelogic quality management user reviews and ratings written by amplelogic quality management customers, and evaluate pricing and features. Amplelogic: document management system on cphi online, the leading search engine in the global pharma industry. Read the latest, in depth amplelogic reviews from real users verified by gartner peer insights, and choose your business software with confidence. Decreases risk of product recalls, improves product quality and safety. improve processes & solve process problems, and provide new preventive actions. easily configurable to meet your specific process and automation needs.
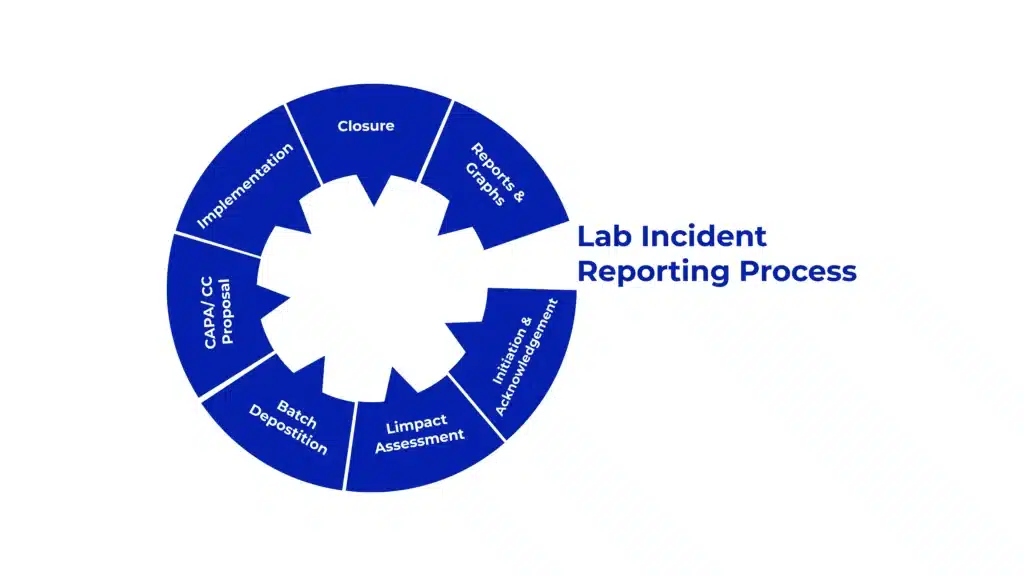
Lab Incident Reporting Software Uae 2025 Amplelogic Read the latest, in depth amplelogic reviews from real users verified by gartner peer insights, and choose your business software with confidence. Decreases risk of product recalls, improves product quality and safety. improve processes & solve process problems, and provide new preventive actions. easily configurable to meet your specific process and automation needs. The qms is 21 cfr part 11 and eu annex 11 compliant and allows organizations to automate paper based processes and integrate them on a single platform for increased transparency. it is flexible to adapt to various regulatory standards and meets the needs of diverse customers to reduce quality costs. we take content rights seriously. Amplelogic lab incident reporting (lir) software provides a comprehensive solution for incident management in pharmaceutical, biotech, healthcare, and life science organizations. Ai powered pharma compliance software for qms, lims & dms. ensure gxp and 21 cfr part 11 compliance, improve quality, and streamline operations. Amplelogic lims is an integrated solution for managing lab data, sample records, test tracking, customer services, and invoices. it ensures end to end traceability of lab workflows from sample receipt to result reporting, enabling faster turnaround times and regulatory compliance.

Lab Incident Reporting Software Uae 2025 Amplelogic The qms is 21 cfr part 11 and eu annex 11 compliant and allows organizations to automate paper based processes and integrate them on a single platform for increased transparency. it is flexible to adapt to various regulatory standards and meets the needs of diverse customers to reduce quality costs. we take content rights seriously. Amplelogic lab incident reporting (lir) software provides a comprehensive solution for incident management in pharmaceutical, biotech, healthcare, and life science organizations. Ai powered pharma compliance software for qms, lims & dms. ensure gxp and 21 cfr part 11 compliance, improve quality, and streamline operations. Amplelogic lims is an integrated solution for managing lab data, sample records, test tracking, customer services, and invoices. it ensures end to end traceability of lab workflows from sample receipt to result reporting, enabling faster turnaround times and regulatory compliance.

Smart Lab Report Software Redefining Laboratory Reporting Ai powered pharma compliance software for qms, lims & dms. ensure gxp and 21 cfr part 11 compliance, improve quality, and streamline operations. Amplelogic lims is an integrated solution for managing lab data, sample records, test tracking, customer services, and invoices. it ensures end to end traceability of lab workflows from sample receipt to result reporting, enabling faster turnaround times and regulatory compliance.
Comments are closed.