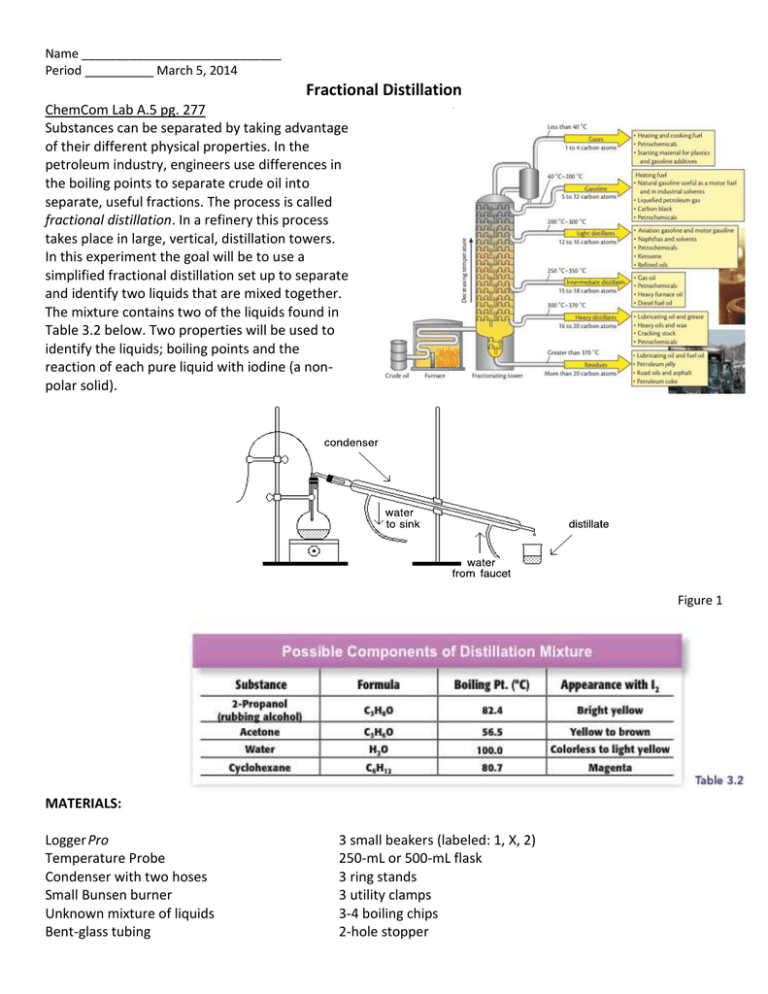
Lab 5 Fractional Distillation

Group 5 Fractional Distillation Pdf It is assumed that readers have previously performed a simple distillation, so in this section are described differences between simple and fractional distillation. The purpose of this experiment was to assess the effectiveness of fractional distillation in the separation of ethanol water (20%:80%) and cyclohexane toluene (50:50%).

Fractional Distillation Lab By Sickie Science Tpt This document describes the process of fractional distillation. it explains that fractional distillation uses a fractionating column to more efficiently separate liquid mixtures than simple distillation. In this experiment fractional distillation will be performed in order to separate ethyl acetate from butyl acetate. also, a comparison should be discussed with the simple distillation experiment performed previously. assemble the apparatus as shown in the diagram. Lab report on fractional distillation experiment, including data, analysis, and conclusion. covers separation and identification of substances. Position and clamp the first 15 ml centrifuge tube (labeled fraction a) below the vacuum adapter such that liquid flowing out of the adapter will flow into the falcon tube.
Fractional Distillation Lab Worksheet Lab report on fractional distillation experiment, including data, analysis, and conclusion. covers separation and identification of substances. Position and clamp the first 15 ml centrifuge tube (labeled fraction a) below the vacuum adapter such that liquid flowing out of the adapter will flow into the falcon tube. Fractional distillation leads to a better separation than simple distillation because the glass beads in the fractionating column provide "theoretical plates" on which the vapors can condense and then re evaporate, and re condense, essentially distilling the compound many times over. Fractional distillation is used for both oil refining and purification of reagents and products. fractional distillation is used in oil refineries (figure 5.41) to separate the complex mixture into fractions that contain similar boiling points and therefore similar molecular weights and properties. From the video, sketch the set up for a fractional distillation. be sure to label the following: heating mantle, round bottom flask, boiling chips, fractionating column, three way adaptor, thermometer adaptor, thermometer, condenser, flow director, keck clamps, and collecting (receiver) flask vial. The lab focused on the separation and purification of organic liquids through various distillation methods, including simple and fractional distillation. the experiments compared the effectiveness of these methods on pure hexane and a binary mixture of hexane and toluene.

Fractional Distillation Diagram Quizlet Fractional distillation leads to a better separation than simple distillation because the glass beads in the fractionating column provide "theoretical plates" on which the vapors can condense and then re evaporate, and re condense, essentially distilling the compound many times over. Fractional distillation is used for both oil refining and purification of reagents and products. fractional distillation is used in oil refineries (figure 5.41) to separate the complex mixture into fractions that contain similar boiling points and therefore similar molecular weights and properties. From the video, sketch the set up for a fractional distillation. be sure to label the following: heating mantle, round bottom flask, boiling chips, fractionating column, three way adaptor, thermometer adaptor, thermometer, condenser, flow director, keck clamps, and collecting (receiver) flask vial. The lab focused on the separation and purification of organic liquids through various distillation methods, including simple and fractional distillation. the experiments compared the effectiveness of these methods on pure hexane and a binary mixture of hexane and toluene.

Fractional Distillation Nebraska Scientific From the video, sketch the set up for a fractional distillation. be sure to label the following: heating mantle, round bottom flask, boiling chips, fractionating column, three way adaptor, thermometer adaptor, thermometer, condenser, flow director, keck clamps, and collecting (receiver) flask vial. The lab focused on the separation and purification of organic liquids through various distillation methods, including simple and fractional distillation. the experiments compared the effectiveness of these methods on pure hexane and a binary mixture of hexane and toluene.
Comments are closed.