Iso 13485 Test Method Validation Headsultra

Iso 13485 2016 Pdf Verification And Validation Medical Device Test method validation: the test method (e.g., test parameters, acceptance criteria, and procedures) must be validated to ensure it is repeatable, reproducible, and capable of detecting leaks reliably. With the transition of many medical device companies to iso 13485:2016 “medical devices – quality management systems – requirements for regulatory purposes”, it is a challenge for the industry to obtain suitably qualified software validation engineers to fulfil its requirements.
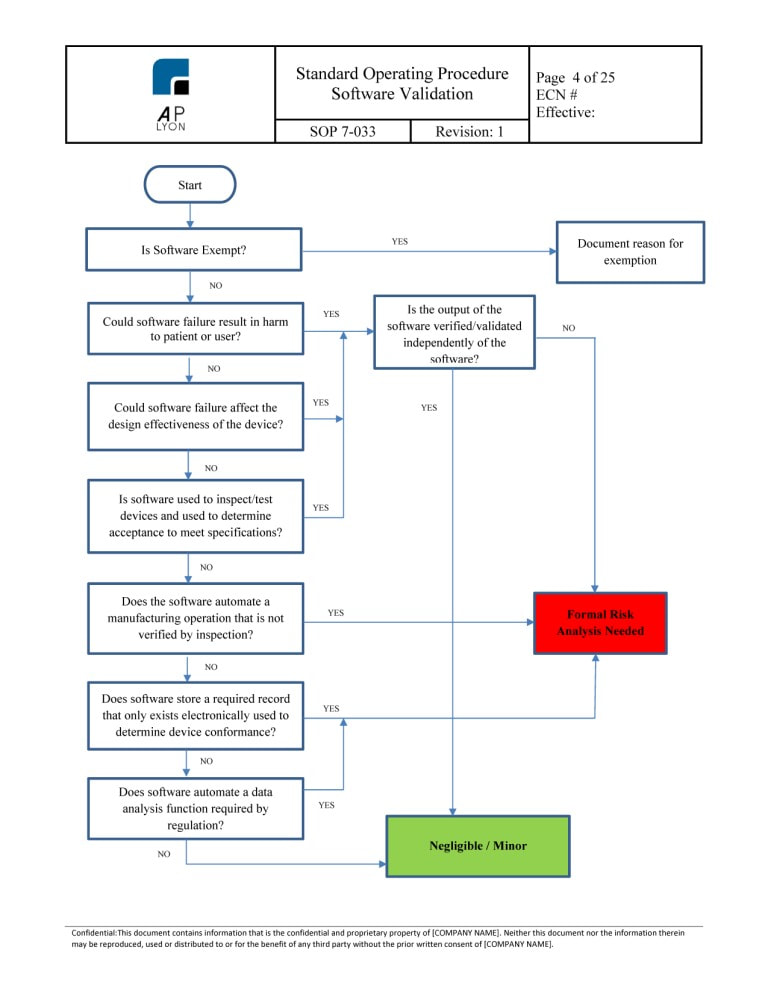
Iso 13485 Test Method Validation Headsultra Our test method validation (tmv) guides provide a clear and structured approach to ensure your test methods for sterile medical packaging are accurate, repeatable, and compliant. pre built templates, checklists, and video tutorials help you streamline validation and avoid common pitfalls. Discover expert medical device process validation with operon strategist. get compliant iq, oq, pq protocols, fda and iso 13485 guidance, and end to end validation consulting for safe, effective devices. Within iso 13485, sterilization and validation processes are critical requirements for the safety and effectiveness of medical devices. this guide aims to clarify the steps that manufacturers and quality management teams frequently encounter in practice. The test method validation process starts with the inspection requirement, capturing and classifying the test methods based on the test and how it is carried out in the manufacturing facility.

Iso 13485 Test Method Validation Headsultra Within iso 13485, sterilization and validation processes are critical requirements for the safety and effectiveness of medical devices. this guide aims to clarify the steps that manufacturers and quality management teams frequently encounter in practice. The test method validation process starts with the inspection requirement, capturing and classifying the test methods based on the test and how it is carried out in the manufacturing facility. There are several steps that you need to go through to achieve iso 13485 certification. the below details each of these steps. after you sign the contract with us, a bsi regional planner will contact you to arrange the review, and you will be assigned a client manager. Learn how test method validation improves data integrity, supports compliance, and reduces risk in medical device development. This course provides regulatory quality systems professionals, manufacturing engineers, and process development engineers with the knowledge and skills needed to comply with the test method validation requirements of the fda's quality system regulation, eu mdr 2017 745, iso 13485 and other regulatory requirements on how to implement an. This piece outlines a step by step approach to iso 13485 software validation that meets regulatory requirements while you retain control of your development process.
Iso 13485 Test Method Validation Headsultra There are several steps that you need to go through to achieve iso 13485 certification. the below details each of these steps. after you sign the contract with us, a bsi regional planner will contact you to arrange the review, and you will be assigned a client manager. Learn how test method validation improves data integrity, supports compliance, and reduces risk in medical device development. This course provides regulatory quality systems professionals, manufacturing engineers, and process development engineers with the knowledge and skills needed to comply with the test method validation requirements of the fda's quality system regulation, eu mdr 2017 745, iso 13485 and other regulatory requirements on how to implement an. This piece outlines a step by step approach to iso 13485 software validation that meets regulatory requirements while you retain control of your development process.

Iso 13485 Test Method Validation Volblind This course provides regulatory quality systems professionals, manufacturing engineers, and process development engineers with the knowledge and skills needed to comply with the test method validation requirements of the fda's quality system regulation, eu mdr 2017 745, iso 13485 and other regulatory requirements on how to implement an. This piece outlines a step by step approach to iso 13485 software validation that meets regulatory requirements while you retain control of your development process.

Iso 13485 Test Method Validation Volblind
Comments are closed.