Iso 13485 Ggm
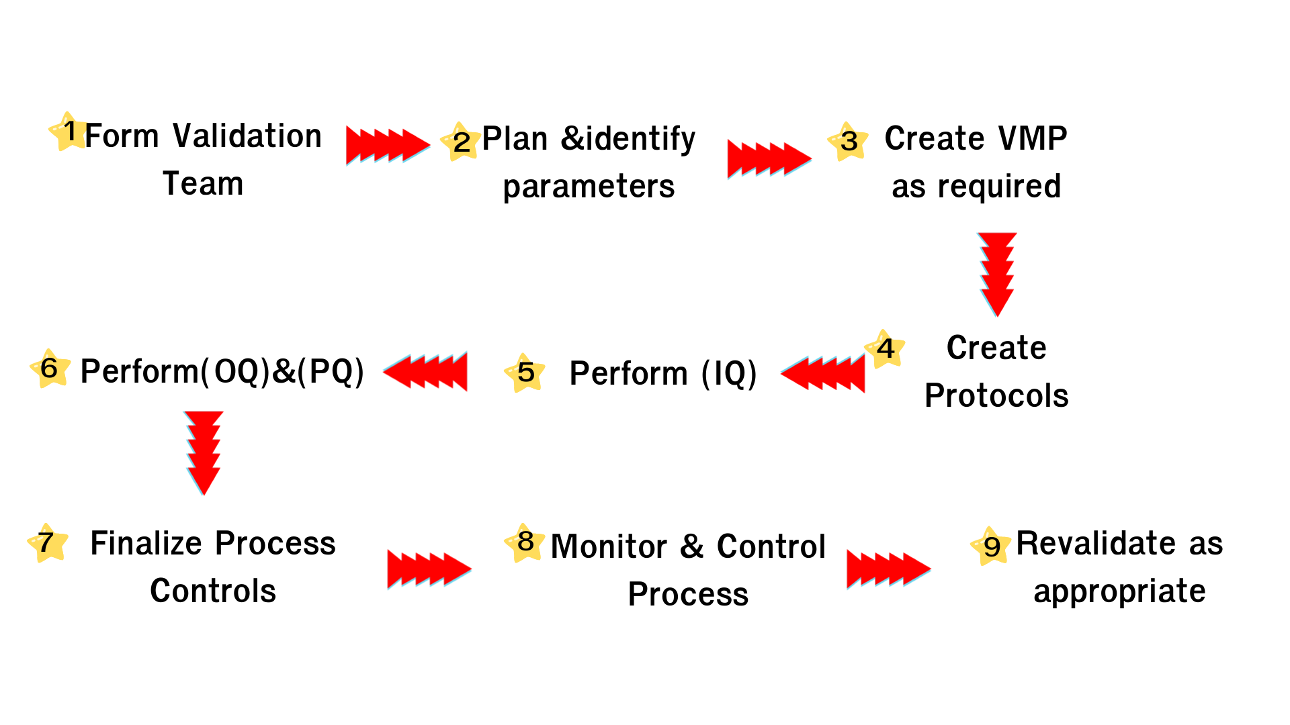
Iso 13485 Ggm Popular standards iso 13485 medical devices safety and quality are non negotiable in the medical devices industry, that’s why we developed iso 13485. regulatory requirements are increasingly stringent throughout every step of a product’s life cycle, including service and delivery. The qmsr incorporates by reference the international standard, iso 13485:2016, medical devices quality management systems requirements for regulatory purposes and clause 3 of iso 9000:2015.

International Standard Ggm Panduan ini dirancang untuk memandu anda melalui langkah langkah penting dalam membangun dan memelihara qms yang sesuai dengan iso 13485 dan mendapatkan sertifikasi. Understand the key differences between gmp and iso 13485 standards. compare their goals, requirements, and how they apply to medical device manufacturing. According to the official definition by the international organization for standardization (iso), iso 13485 specifies requirements for a quality management system where an organization needs to demonstrate its ability to provide medical devices and related services that consistently meet customer and applicable regulatory requirements. This guide will walk you through the globally harmonized standard for medical devices iso 13485 quality management system.

Contoh Iso 13485 Pdf According to the official definition by the international organization for standardization (iso), iso 13485 specifies requirements for a quality management system where an organization needs to demonstrate its ability to provide medical devices and related services that consistently meet customer and applicable regulatory requirements. This guide will walk you through the globally harmonized standard for medical devices iso 13485 quality management system. This guide is crafted to demystify the intricacies of iso 13485, offering crucial insights to elevate your product quality and boost customer satisfaction. Iso 13485:2016 is an internationally recognized standard that outlines the requirements for a quality management system (qms) specifically developed for organizations involved in the design, development, production, installation, and servicing of medical devices. Iso 13485 is a globally recognized medical device certification standard to help you demonstrate that your management system can meet product and regulatory requirements. Learn about iso 13485 quality management system for medical devices and bsi group's role in supporting regulatory compliance. we set the standards for the world's biggest companies.

Iso 13485 Lembaga Sertifikasi Terpercaya This guide is crafted to demystify the intricacies of iso 13485, offering crucial insights to elevate your product quality and boost customer satisfaction. Iso 13485:2016 is an internationally recognized standard that outlines the requirements for a quality management system (qms) specifically developed for organizations involved in the design, development, production, installation, and servicing of medical devices. Iso 13485 is a globally recognized medical device certification standard to help you demonstrate that your management system can meet product and regulatory requirements. Learn about iso 13485 quality management system for medical devices and bsi group's role in supporting regulatory compliance. we set the standards for the world's biggest companies.
Comments are closed.