Iqvia Regulatory Intelligence
Regulatory Intelligence Iqvia regulatory intelligence provides instant access to regulatory insights and real time updates from national authorities across the globe, helping you to avoid the resource intensive process of gathering, organizing, and sharing important regulatory information. Iqvia regulatory intelligence provides insights and solutions to navigate regulatory challenges, ensuring compliance and enabling smarter decision making in healthcare and life sciences.

Iqvia Regulatory Intelligence Iqvia With iqvia regulatory intelligence, you can access regulatory information for human drugs and biologics, medical devices and ivds in over 100 countries, regions, and international organizations, all in a newly designed platform, you can get the latest regulatory insights and real time updates you need all in one place. Providing you with regulatory strategy, labeling, publishing, and intelligence solutions supported by our global expertise, data and technology, from early drug development through submissions and post registration. navigate changing regulatory demands with greater accuracy, agility and efficiency. let us help you where you need it most. Iqvia regulatory intelligence provides access to regulatory requirements for drugs, biologics, medical devices and ivds in over 110 countries and regions. you can customize your database with various tools and request more information or contact an iqvia expert. Log in to regulatory intelligence email address * continue for further support, contact iqvia regulatory intelligence.

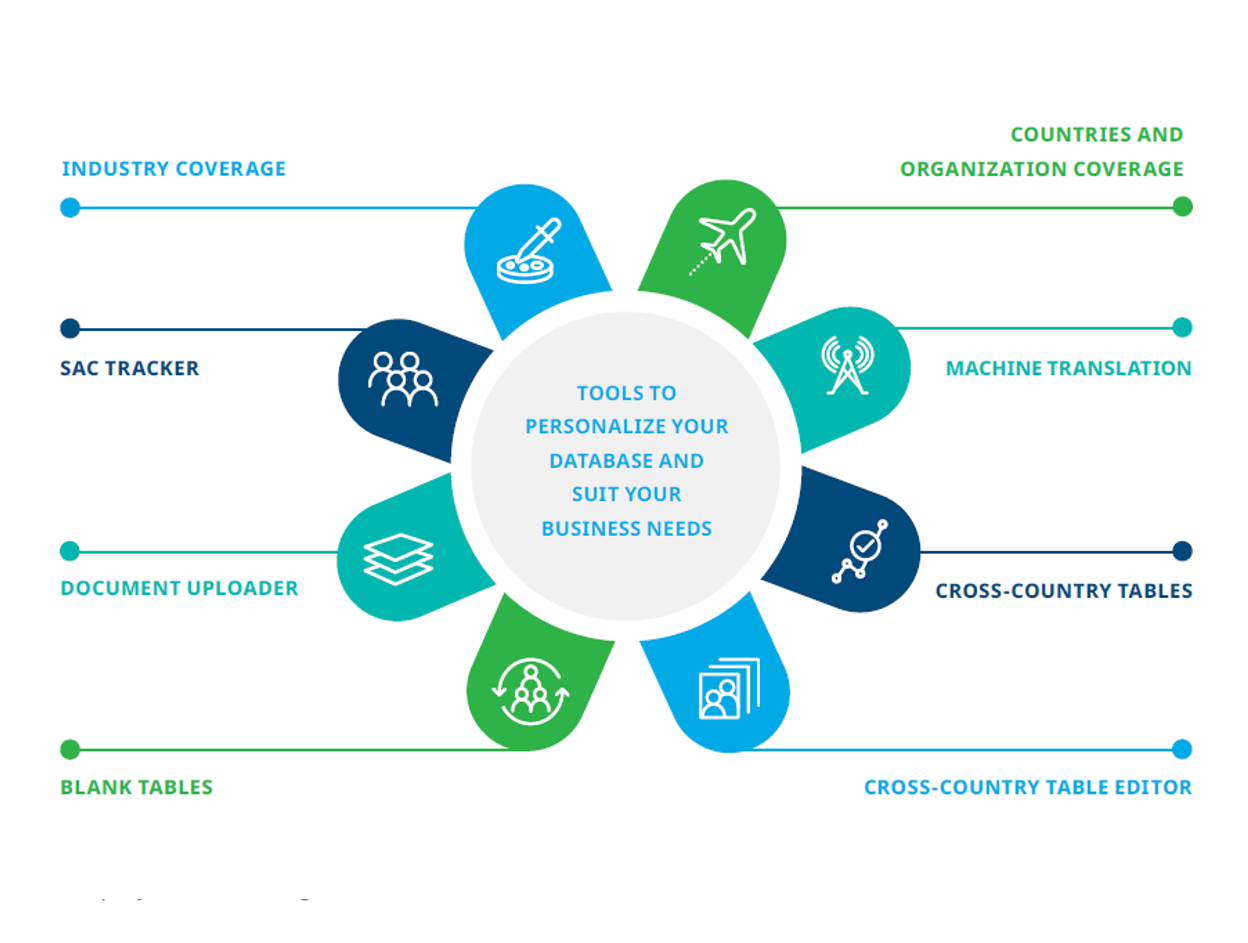
Iqvia Regulatory Intelligence Iqvia Iqvia regulatory intelligence provides access to regulatory requirements for drugs, biologics, medical devices and ivds in over 110 countries and regions. you can customize your database with various tools and request more information or contact an iqvia expert. Log in to regulatory intelligence email address * continue for further support, contact iqvia regulatory intelligence. Iqvia regulatory intelligence provides access to regulatory insights and updates for human drugs & biologics and medical devices & ivds, for around 110 countries and regions. you can customize your database with various tools and enhance your coverage in areas such as cell and gene therapy, samd, safety, clinical trials and cross country. Iqvia's regulatory intelligence provides insights and solutions for regulatory compliance and strategy in the pharmaceutical and healthcare industries. Tarius is a dedicated platform that provides access to regulatory requirements for the healthcare industry on human drugs, biologics, medical devices, and ivds. it covers more than 100 countries, regions and international organizations worldwide. Streamline your regulatory compliance systems and processes with our new end to end regulatory technologies, advisory and tech enabled services. free your team from labor intensive tasks and maintenance to focus more on delivering valuable products to market.

Iqvia Regulatory Intelligence Iqvia Iqvia regulatory intelligence provides access to regulatory insights and updates for human drugs & biologics and medical devices & ivds, for around 110 countries and regions. you can customize your database with various tools and enhance your coverage in areas such as cell and gene therapy, samd, safety, clinical trials and cross country. Iqvia's regulatory intelligence provides insights and solutions for regulatory compliance and strategy in the pharmaceutical and healthcare industries. Tarius is a dedicated platform that provides access to regulatory requirements for the healthcare industry on human drugs, biologics, medical devices, and ivds. it covers more than 100 countries, regions and international organizations worldwide. Streamline your regulatory compliance systems and processes with our new end to end regulatory technologies, advisory and tech enabled services. free your team from labor intensive tasks and maintenance to focus more on delivering valuable products to market.

Iqvia Regulatory Intelligence Iqvia Tarius is a dedicated platform that provides access to regulatory requirements for the healthcare industry on human drugs, biologics, medical devices, and ivds. it covers more than 100 countries, regions and international organizations worldwide. Streamline your regulatory compliance systems and processes with our new end to end regulatory technologies, advisory and tech enabled services. free your team from labor intensive tasks and maintenance to focus more on delivering valuable products to market.

Iqvia Regulatory Intelligence Iqvia
Comments are closed.