Ionic Strength Definition Effect Debye Huckel

Debye Huckel Theory Pdf The most significant aspect of equation 25.6.1 is the prediction that the mean activity coefficient is a function of ionic strength rather than the electrolyte concentration. Activity coefficients are themselves functions of concentration, since the amount of inter ionic interaction increases as the concentration of the electrolyte increases. debye and hückel developed a theory with which single ion activity coefficients could be calculated.

Debye Huckel Limiting Law Ionic Strength And Activity Coefficient Understand ionic strength, its calculation, significance, and impact on chemical equilibrium explained in simple terms. This equation can be solved on computer and is used for biophysical modeling, for example, to study the ionic atmosphere around dna, and the effects of salt on the electrostatic potentials of proteins. Ionic strength (i): a measure of the total concentration of ions in the solution. the debye hückel limiting law is expressed as: ln (γ i) = a·z i2 √i. where a is a constant dependent on the temperature and the dielectric constant of the solvent, and z i is the charge of the ion. In the next unit we will continue the discussion on debye hückel theory in terms of debye hückel limiting law, discuss its significance and take up the experimental determination of activity coefficients.
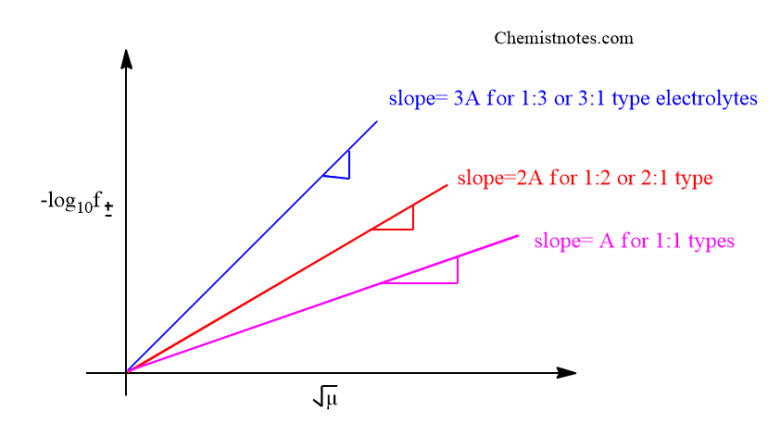
Debye Huckel Limiting Law Ionic Strength And Activity Coefficient Ionic strength (i): a measure of the total concentration of ions in the solution. the debye hückel limiting law is expressed as: ln (γ i) = a·z i2 √i. where a is a constant dependent on the temperature and the dielectric constant of the solvent, and z i is the charge of the ion. In the next unit we will continue the discussion on debye hückel theory in terms of debye hückel limiting law, discuss its significance and take up the experimental determination of activity coefficients. For solutions of relatively low ionic strength, the activity coefficient of an ion can be estimated by the debye–hückel theory. this uses a model with each ion surrounded by an atmosphere of ions of the same charge, but which is opposite to that of the central ion. Ionic strength of a solution can be defined as the total concentration of ions present in the solution. in another word, ionic strength measures the concentration of ionic atmosphere in a solution. It explains the interactions between ions in a solution and introduces the debye huckel limiting law, which describes the relationship between ionic strength and activity coefficients. the document also covers the mathematical derivations and assumptions underlying the theory. Ionic strength affects the activity of ions, which is important for understanding chemical equilibria and reaction rates in biological systems. the debye hückel theory provides a mathematical model for describing the effects of ionic strength on the behavior of ions and charged particles.
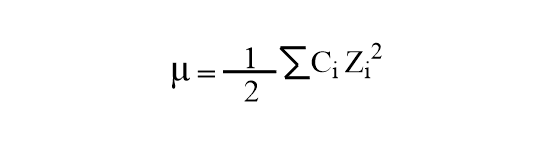
Debye Huckel Limiting Law Ionic Strength And Activity Coefficient For solutions of relatively low ionic strength, the activity coefficient of an ion can be estimated by the debye–hückel theory. this uses a model with each ion surrounded by an atmosphere of ions of the same charge, but which is opposite to that of the central ion. Ionic strength of a solution can be defined as the total concentration of ions present in the solution. in another word, ionic strength measures the concentration of ionic atmosphere in a solution. It explains the interactions between ions in a solution and introduces the debye huckel limiting law, which describes the relationship between ionic strength and activity coefficients. the document also covers the mathematical derivations and assumptions underlying the theory. Ionic strength affects the activity of ions, which is important for understanding chemical equilibria and reaction rates in biological systems. the debye hückel theory provides a mathematical model for describing the effects of ionic strength on the behavior of ions and charged particles.

Debye Huckel Limiting Law Ionic Strength And Activity Coefficient It explains the interactions between ions in a solution and introduces the debye huckel limiting law, which describes the relationship between ionic strength and activity coefficients. the document also covers the mathematical derivations and assumptions underlying the theory. Ionic strength affects the activity of ions, which is important for understanding chemical equilibria and reaction rates in biological systems. the debye hückel theory provides a mathematical model for describing the effects of ionic strength on the behavior of ions and charged particles.
Comments are closed.