Ionic Compounds And Bonding
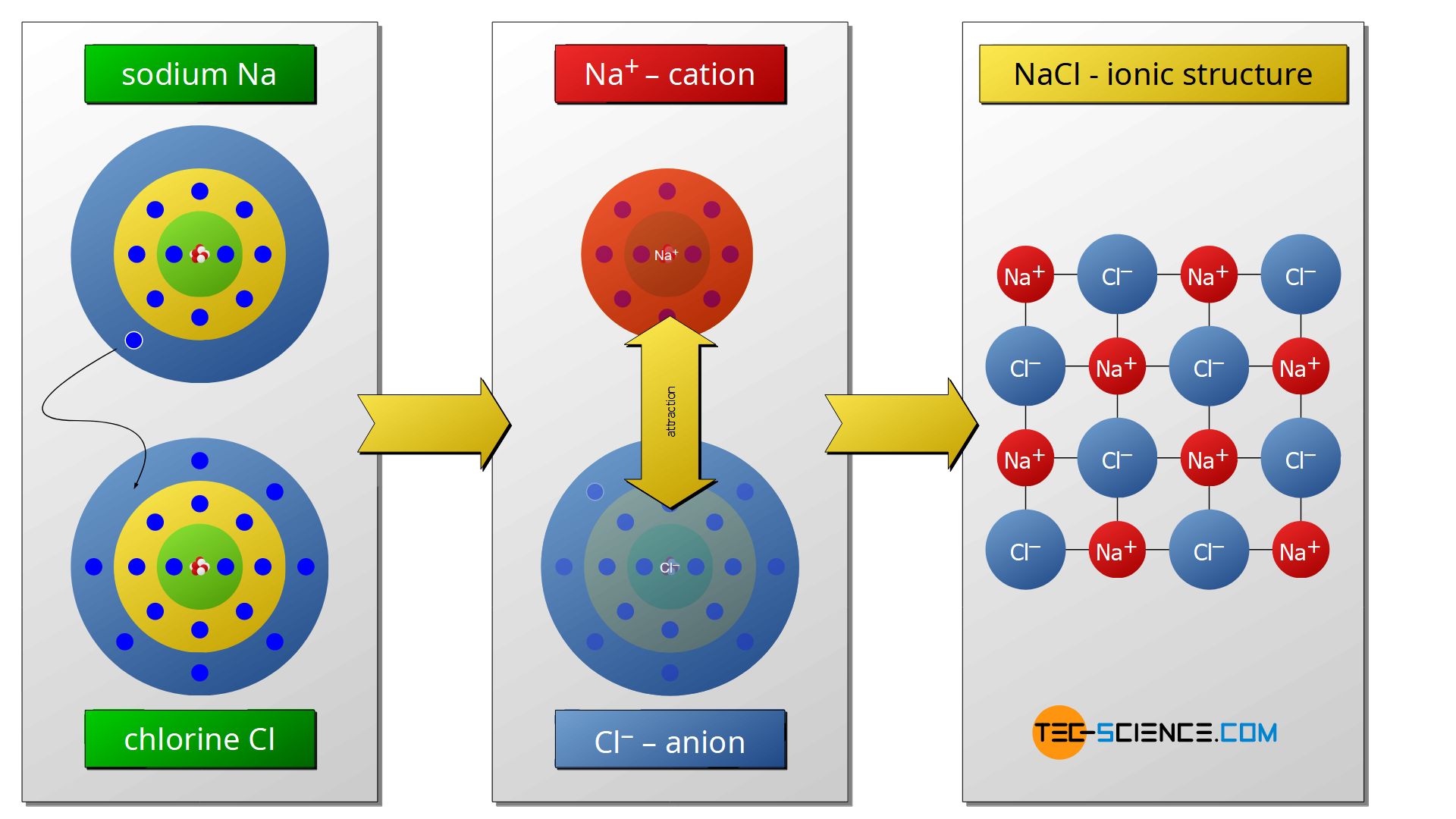
Ionic Bonding Compounds Exles Infoupdate Org An ionic bond, also known as an electrovalent bond, is a type of chemical bond formed due to the electrostatic attraction between oppositely charged ions in an ionic compound. This lesson shows how cations and anions combine in specific ratios to form electrically neutral ionic compounds, and how ionic bonds shape their characteristic crystal lattice structures.
Ionic Bonding Compounds Exles Infoupdate Org Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom. learn more about ionic bonds in this article. This page explains ionic bonds and compounds through electrostatic forces, detailing how oppositely charged ions attract. using sodium chloride (nacl) as an example, it describes the electron transfer that forms na⁺ and cl⁻ ions. Ionic compound is a chemical compound composed of cations and anions held together by ionic bonds. ionic compounds form when atoms transfer electrons to achieve a stable electron configuration, typically forming an extended crystal lattice of alternating ions. The strength of the ionic bonds depends on the charge on the ions. ions with higher charge will have stronger forces between them, so will need more energy in order to overcome these forces.

Ionic Bonding Compounds Exles Infoupdate Org Ionic compound is a chemical compound composed of cations and anions held together by ionic bonds. ionic compounds form when atoms transfer electrons to achieve a stable electron configuration, typically forming an extended crystal lattice of alternating ions. The strength of the ionic bonds depends on the charge on the ions. ions with higher charge will have stronger forces between them, so will need more energy in order to overcome these forces. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: electrostatic forces of attraction between oppositely charged cations and anions. Ionic compounds contain ionic bonds. an ionic bond is formed when there is a large electronegativity difference between the elements participating in the bond. the greater the difference, the stronger the attraction between the positive ion (cation) and negative ion (anion). Ionic bonding occurs in compounds composed of strongly electropositive elements (metals) and strongly electronegative elements (nonmetals). the alkali halides (nacl, lif, etc.) are the most unambiguous examples of ionically bonded solids. When focusing on ionic bonding, it is essential to understand that ionic compounds, also known as ionic salts, consist of a positive ion (cation) and a negative ion (anion). the attraction between these opposing charges is what enables the formation of ionic compounds.

Ionic Bonding And Ionic Compounds Chemistry Worksheet Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: electrostatic forces of attraction between oppositely charged cations and anions. Ionic compounds contain ionic bonds. an ionic bond is formed when there is a large electronegativity difference between the elements participating in the bond. the greater the difference, the stronger the attraction between the positive ion (cation) and negative ion (anion). Ionic bonding occurs in compounds composed of strongly electropositive elements (metals) and strongly electronegative elements (nonmetals). the alkali halides (nacl, lif, etc.) are the most unambiguous examples of ionically bonded solids. When focusing on ionic bonding, it is essential to understand that ionic compounds, also known as ionic salts, consist of a positive ion (cation) and a negative ion (anion). the attraction between these opposing charges is what enables the formation of ionic compounds.
Comments are closed.