Ionic Bonding Notes

Ionic Bonding Notes Pdf Ionic bonds form when one atom gives up electrons, and another atom takes them. this happens because atoms want to have a full outer shell of electrons (like noble gases) to become stable. a metal atom (like sodium, na) loses its outer electron(s) because it’s easier to lose than gain. Use our revision notes to learn about ionic bonding for your gcse chemistry exam. define an ion and draw dot and cross diagrams. learn more.

Ionic Bonding Ocr Gateway Gcse Chemistry Revision Notes 40 Off An ionic bond is formed when electrons are transferred from a metal to a non metal, forming an ionic compound. the compound is held together by the electrostatic attraction between the positively charged metal ions and negatively charged non metal ions. It is important to know how to and covert ionic formula of an ionic compound to a name and vice versa and this will help to apply solve chemical problems involving ionic compounds. Understand, in simple terms, the concept of electronegativity and apply it to explain the properties of molecules such as bond polarity and the dipole moments of molecules. Learn about ionic and covalent bonding, how metals react to form ionic compounds and how this affects their properties.

Ionic Bonding Notes By The Science Hedgehog Tpt Understand, in simple terms, the concept of electronegativity and apply it to explain the properties of molecules such as bond polarity and the dipole moments of molecules. Learn about ionic and covalent bonding, how metals react to form ionic compounds and how this affects their properties. Ionic bonds are characterized by the complete transfer of electrons from one atom to another, resulting in the formation of two charged particles known as ions, which are held together with the help of electrostatic forces. An ionic bond is the electrostatic force of attraction between two oppositely charged ions (called anions and cations). ionic bonds can occur between metals and non metals. Get the ionic bond definition and examples in chemistry. learn which types of atoms participate in ionic bonding. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: electrostatic forces of attraction between oppositely charged cations and anions. the properties of ionic compounds shed some light on the nature of ionic bonds.
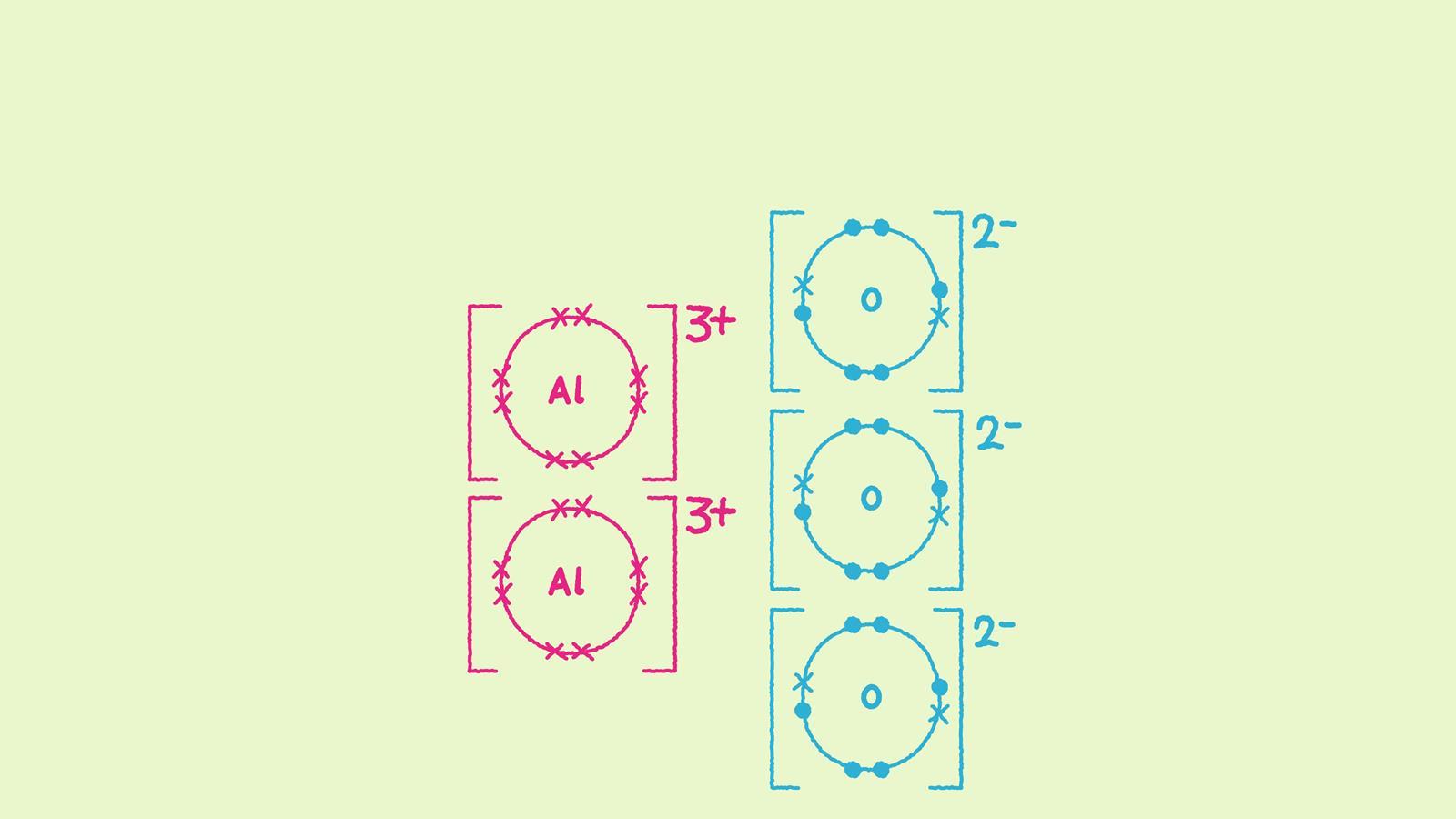
Ionic Bonding Diagram Ionic bonds are characterized by the complete transfer of electrons from one atom to another, resulting in the formation of two charged particles known as ions, which are held together with the help of electrostatic forces. An ionic bond is the electrostatic force of attraction between two oppositely charged ions (called anions and cations). ionic bonds can occur between metals and non metals. Get the ionic bond definition and examples in chemistry. learn which types of atoms participate in ionic bonding. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: electrostatic forces of attraction between oppositely charged cations and anions. the properties of ionic compounds shed some light on the nature of ionic bonds.

Ionic Bonding Notes Pdf Ion Chemical Compounds Get the ionic bond definition and examples in chemistry. learn which types of atoms participate in ionic bonding. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: electrostatic forces of attraction between oppositely charged cations and anions. the properties of ionic compounds shed some light on the nature of ionic bonds.
Comments are closed.