Ionic Bonding Introduction

Ionic Bonding Introduction Worksheet By Chempro Tpt Ionic bonds form when one atom gives up electrons, and another atom takes them. this happens because atoms want to have a full outer shell of electrons (like noble gases) to become stable. a metal atom (like sodium, na) loses its outer electron(s) because it’s easier to lose than gain. Ionic bonding is a type of chemical bonding that involves the electrostatic attraction between oppositely charged ions, or between two atoms with sharply different electronegativities, [1] and is the primary interaction occurring in ionic compounds.

Ionic Bonding Introduction Worksheet By Chempro Tpt An ionic bond is an electrostatic attraction between charged particles. recall from chapter 2 that protons, which are positive, and electrons, which are negative, are attracted to one another. larger charged particles, which are called ions, behave analogously. What is an ionic bond? an ionic bond, also known as an electrovalent bond, is a type of chemical bond formed due to the electrostatic attraction between oppositely charged ions in an ionic compound. An ionic bond is a bond in which an atom donates one or more electrons to another atom to produce ions (cations and anions). it is a form of chemical bonding that involves electrostatic attractions between oppositely charged ions or between two atoms with sharply dissimilar electronegativities. Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom. learn more about ionic bonds in this article.
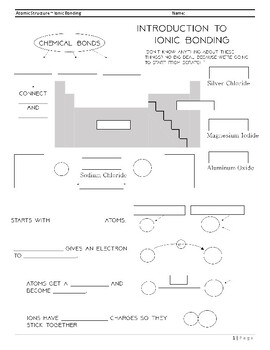
Ionic Bonding Introduction Worksheet By Chempro Tpt An ionic bond is a bond in which an atom donates one or more electrons to another atom to produce ions (cations and anions). it is a form of chemical bonding that involves electrostatic attractions between oppositely charged ions or between two atoms with sharply dissimilar electronegativities. Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom. learn more about ionic bonds in this article. Therefore an ionic bond is simply the electrostatic attraction between oppositely charged ions. ionic compounds consist of giant structure of ions called an ionic lattice. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: electrostatic forces of attraction between oppositely charged cations and anions. the properties of ionic compounds shed some light on the nature of ionic bonds. Ionic bonding is a type of chemical bond in which valence electrons are lost from one atom and gained by another. this exchange results in a more stable, noble gas electronic configuration for both atoms involved. Ionic bonding is one of the main types of chemical bonding, together with covalent bonding and metallic bonding. an ionic bond is when one atom donates its valence electron to another atom, increasing the stability of both atoms.
Comments are closed.