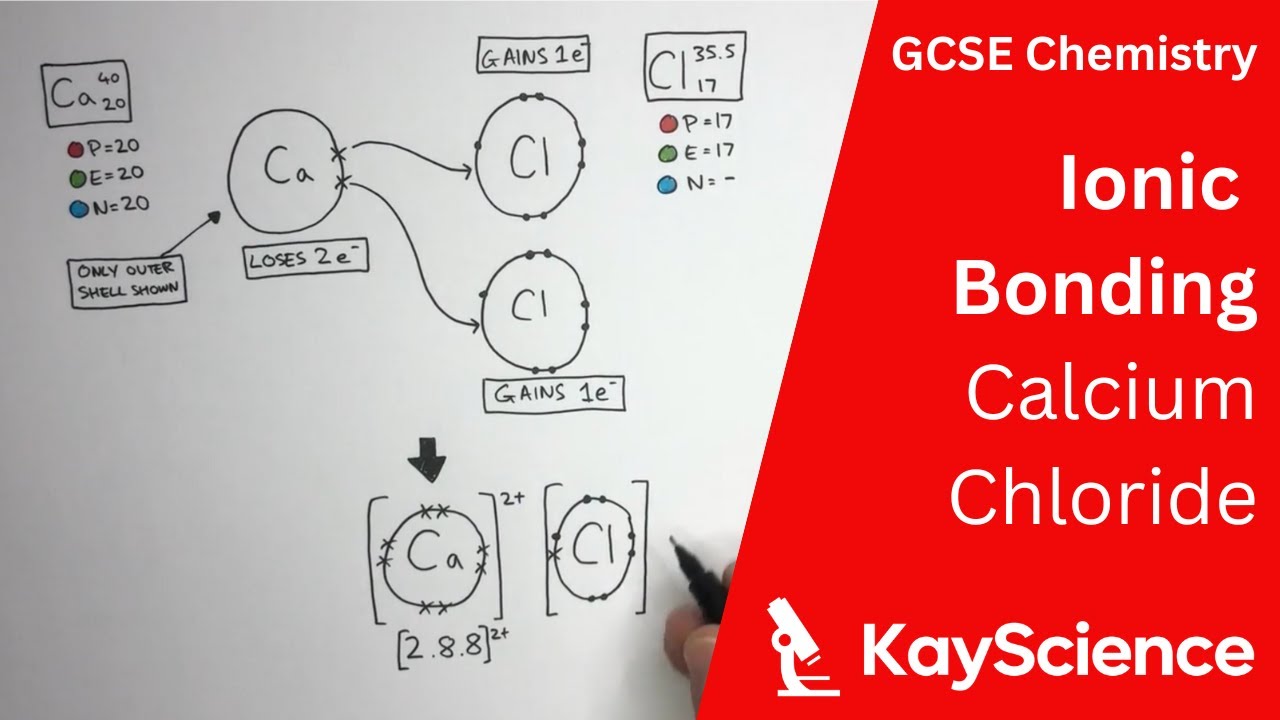
Ionic Bonding Explained Formation And Bonds
Ionic Bonding Of Magnesium Oxide Explained Gcse Chemistry 41 Off What is an ionic bond? an ionic bond, also known as an electrovalent bond, is a type of chemical bond formed due to the electrostatic attraction between oppositely charged ions in an ionic compound. In ionic bonding, the atoms are bound by the attraction of oppositely charged ions, whereas, in covalent bonding, atoms are bound by sharing electrons to attain stable electron configurations.

Understanding Ionic Bonds Definition And Formation Explained Learn Ionic bonding is the attraction between positively and negatively charged ions. these oppositely charged ions attract each other to form ionic networks (or lattices). electrostatics explains why this happens: opposite charges attract and like charges repel. This lesson shows how cations and anions combine in specific ratios to form electrically neutral ionic compounds, and how ionic bonds shape their characteristic crystal lattice structures. The formation of ionic compounds binary ionic compounds are composed of just two elements: a metal (which forms the cations) and a nonmetal (which forms the anions). for example, [latex]\ce {nacl} [ latex] is a binary ionic compound. we can think about the formation of such compounds in terms of the periodic properties of the elements. Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom. learn more about ionic bonds in this article.

Ionic Bonding Ocr Gateway Gcse Chemistry Revision Notes 40 Off The formation of ionic compounds binary ionic compounds are composed of just two elements: a metal (which forms the cations) and a nonmetal (which forms the anions). for example, [latex]\ce {nacl} [ latex] is a binary ionic compound. we can think about the formation of such compounds in terms of the periodic properties of the elements. Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom. learn more about ionic bonds in this article. An ionic bond is the electrostatic force that holds ions together in an ionic compound. the strength of the ionic bond is directly dependent upon the quantity of the charges and inversely dependent on the distance between the charged particles. Ionic bonding is defined as a type of chemical bond that occurs when an atom donates one or more electrons to another atom, resulting in the formation of oppositely charged ions that are held together by electrostatic attractions. In this article, the author has explained ionic bonding, conditions favorable for the formation of ionic bond, and factors affecting ionic bond formation. the author has also explained the properties of ionic compounds and examples of ionic compounds. Ionic bonds are formed between a metal cation and a non metal anion due to the electrostatic force that binds ions of opposite charge. this type of bond results from the complete transfer of one or more electrons from one atom to another, leading to the formation of 2 charged particles, called ions.

The Formation And Properties Of Ionic Bonds Explaining The Factors An ionic bond is the electrostatic force that holds ions together in an ionic compound. the strength of the ionic bond is directly dependent upon the quantity of the charges and inversely dependent on the distance between the charged particles. Ionic bonding is defined as a type of chemical bond that occurs when an atom donates one or more electrons to another atom, resulting in the formation of oppositely charged ions that are held together by electrostatic attractions. In this article, the author has explained ionic bonding, conditions favorable for the formation of ionic bond, and factors affecting ionic bond formation. the author has also explained the properties of ionic compounds and examples of ionic compounds. Ionic bonds are formed between a metal cation and a non metal anion due to the electrostatic force that binds ions of opposite charge. this type of bond results from the complete transfer of one or more electrons from one atom to another, leading to the formation of 2 charged particles, called ions.

Ionic Bonding Explained Formation And Bonds In this article, the author has explained ionic bonding, conditions favorable for the formation of ionic bond, and factors affecting ionic bond formation. the author has also explained the properties of ionic compounds and examples of ionic compounds. Ionic bonds are formed between a metal cation and a non metal anion due to the electrostatic force that binds ions of opposite charge. this type of bond results from the complete transfer of one or more electrons from one atom to another, leading to the formation of 2 charged particles, called ions.

Ionic Bonding Gcse Chemistry Explained With Examples
Comments are closed.