Introduction To Galvanic Cells Voltaic Cells

Introduction To Galvanic Cell Pdf Electrode Electrochemistry In lesson 2, we will study a type of electrochemical cell known as a galvanic cell (sometimes referred to as a voltaic cell). a galvanic cell is reliant upon a spontaneous redox reaction. Galvanic cells, also known as voltaic cells, are electrochemical cells in which spontaneous oxidation reduction reactions produce electrical energy.
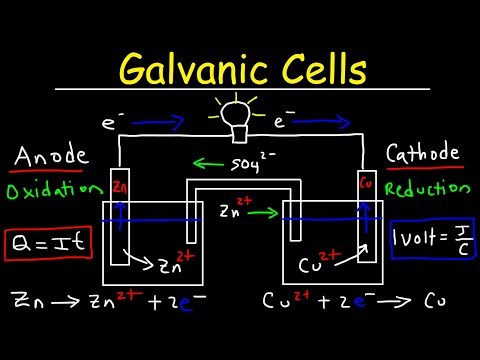
Introduction To Galvanic Cells Voltaic Cells Video Summary And Q A A galvanic cell, also known as a voltaic cell, is a device that can convert chemical energy into electrical energy through spontaneous redox (oxidation reduction) reactions. it is a type of electrochemical cell that is named after italian scientists luigi galvani and alessandro volta. A voltaic (or galvanic) cell is a type of electrochemical cell in which a spontaneous redox reaction generates an electric current. a simple voltaic cell can be constructed using two half cells connected by a salt bridge and an external wire. A galvanic cell uses the energy released from a spontaneous redox reaction to produce an electric current. another common name for galvanic cells is voltaic cells, which is named after another italian physicist, alessandro volta (1745 – 1827), who invented the galvanic (voltaic) cell. In this page, we focus on non rechargeable galvanic cells. use this page to revise the following concepts within galvanic cells: for a galvanic cell an electrochemical cell, also known as a voltaic cell, that converts chemical energy into electrical energy through a spontaneous redox reaction.

Introduction To Galvanic Voltaic Cells Video Lecture General A galvanic cell uses the energy released from a spontaneous redox reaction to produce an electric current. another common name for galvanic cells is voltaic cells, which is named after another italian physicist, alessandro volta (1745 – 1827), who invented the galvanic (voltaic) cell. In this page, we focus on non rechargeable galvanic cells. use this page to revise the following concepts within galvanic cells: for a galvanic cell an electrochemical cell, also known as a voltaic cell, that converts chemical energy into electrical energy through a spontaneous redox reaction. A galvanic cell or voltaic cell, named after the scientists luigi galvani and alessandro volta, respectively, is an electrochemical cell in which an electric current is generated from spontaneous oxidation–reduction reactions. Galvanic cells, also known as voltaic cells, are electrochemical cells in which spontaneous oxidation reduction reactions produce electrical energy. a battery is a galvanic cell. A galvanic cell (or voltaic cell) is a specific type of electrochemical cell in which a spontaneous chemical reaction is harnessed to generate an electric current. A galvanic cell is an electrochemical cell that converts the chemical energy of spontaneous redox reactions into electrical energy. it is also known as a voltaic cell.

Solution Introduction To Galvanic Cells Vs Voltaic Cells Studypool A galvanic cell or voltaic cell, named after the scientists luigi galvani and alessandro volta, respectively, is an electrochemical cell in which an electric current is generated from spontaneous oxidation–reduction reactions. Galvanic cells, also known as voltaic cells, are electrochemical cells in which spontaneous oxidation reduction reactions produce electrical energy. a battery is a galvanic cell. A galvanic cell (or voltaic cell) is a specific type of electrochemical cell in which a spontaneous chemical reaction is harnessed to generate an electric current. A galvanic cell is an electrochemical cell that converts the chemical energy of spontaneous redox reactions into electrical energy. it is also known as a voltaic cell.
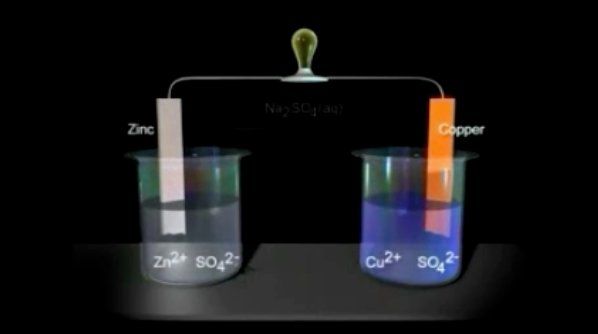
Introduction To Galvanic Voltaic Cells Chemistry Khan Academy A galvanic cell (or voltaic cell) is a specific type of electrochemical cell in which a spontaneous chemical reaction is harnessed to generate an electric current. A galvanic cell is an electrochemical cell that converts the chemical energy of spontaneous redox reactions into electrical energy. it is also known as a voltaic cell.
Comments are closed.