Introduction To Equilibrium

Reversible Reactions And Dynamic Equilibrium Pdf Chemical When a chemical reaction is at equilibrium, any disturbance of the system, such as a change in temperature, or addition or removal of one of the reaction components, will "shift" the composition to a new equilibrium state. This chapter defines equilibrium in mixtures and ideal solutions and introduces the equilibrium constant, its relationship with the gibbs free energy and its dependence on temperature.

Chemical Equilibrium Introduction Chemistryedu Equilibrium: a state of balance in a system, where opposing forces or processes are in balance. at equilibrium, the system’s state does not change over time: d dt f(t) = 0 d d t f (t) = 0. In this lecture, we discuss the nature of chemical equilibrium and of the chemical equilibrium constant. we start to consider how external factors can “push” the equilibrium in one direction or the other. Le châtelier's principle (1884) predicts the behavior of an equilibrium system when changes to its reaction conditions occur. if a dynamic equilibrium is disturbed by changing the conditions, the position of equilibrium moves to partially reverse the change. In a chemical equilibrium, the forward and reverse reactions occur at equal rates, and the concentrations of products and reactants remain constant. if we run a reaction in a closed system so that the products cannot escape, we often find the reaction does not give a 100% yield of products.

Week12 Introduction To Equilibrium 1 Pptx Chemical Equilibrium A Le châtelier's principle (1884) predicts the behavior of an equilibrium system when changes to its reaction conditions occur. if a dynamic equilibrium is disturbed by changing the conditions, the position of equilibrium moves to partially reverse the change. In a chemical equilibrium, the forward and reverse reactions occur at equal rates, and the concentrations of products and reactants remain constant. if we run a reaction in a closed system so that the products cannot escape, we often find the reaction does not give a 100% yield of products. Understand the concept of balance in chemical and physical systems with examples and practice questions. what is equilibrium? equilibrium is a state in which all the forces or processes in a system are balanced. Equilibrium is a fundamental concept in chemistrythat describes the state of a system where the concentrations of reactants and products remain constant over time. At equilibrium, with both the forward and reverse reactions taking place at the same rate, the concentration of every species no longer changes. every reaction has a point in which equilibrium is established. This chapter defines equilibrium in mixtures and ideal solutions and introduces the equilibrium constant, its relationship with the gibbs free energy and its dependence on temperature.
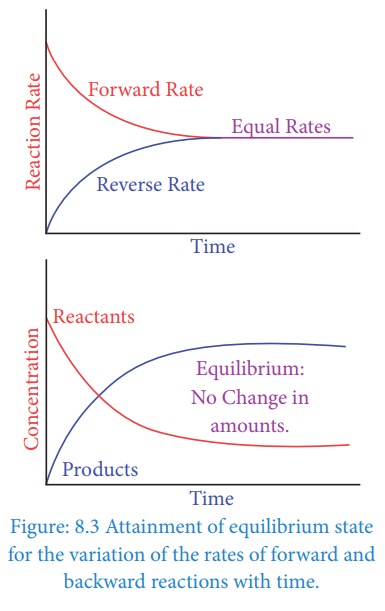
Chemical Equilibrium Understand the concept of balance in chemical and physical systems with examples and practice questions. what is equilibrium? equilibrium is a state in which all the forces or processes in a system are balanced. Equilibrium is a fundamental concept in chemistrythat describes the state of a system where the concentrations of reactants and products remain constant over time. At equilibrium, with both the forward and reverse reactions taking place at the same rate, the concentration of every species no longer changes. every reaction has a point in which equilibrium is established. This chapter defines equilibrium in mixtures and ideal solutions and introduces the equilibrium constant, its relationship with the gibbs free energy and its dependence on temperature.
Comments are closed.