Introduction To Electrochemistry

Introduction To Electrochemistry Pdf Electrochemistry Redox Electrochemistry deals with chemical reactions that produce electricity and the changes associated with the passage of electrical current through matter. the reactions involve electron transfer, and so they are oxidation reduction (or redox) reactions. Electrochemistry deals with oxidation reduction reactions that either produce or utilize electrical energy and electrochemical reactions take place in cells. each cell has two electrodes, conductors through which electrons enter or leave the cell.

Electrochemistry Pdf Redox Electrochemistry Learn the basics of electrochemistry, the science of electric charge transfer across interfaces. find out how electrochemical cells work, how to balance redox reactions, and how to use electrochemistry for energy production and synthesis. Electrochemistry is the study of the relationship between electricity and chemical reactions. the oxidation–reduction reaction that occurs during an electrochemical process consists of two half reactions, one representing the oxidation process and one the reduction process. • electrochemistry is the study of electron charge transfer processes at an electrode solution interface. fe3 e ! fe2 e ! fe3 . cu2 2e ! 2e ! fe 2e ! fe2 cl2. silver and copper electrodes more likely to reduce the species than gold or platinum. a platinum electrode is more likely to oxidise the species than gold, copper or silver. (s) m1 !. This textbook offers original and new approaches to the teaching of electrochemical concepts, principles and applications. throughout the text the authors provide a balanced coverage of the thermodynamic and kinetic processes at the heart of electrochemical systems.

Pdf An Introduction To Electrochemistry An Introduction To Electrochemistry is the study of reactions in which charged particles (ions or electrons) cross the interface between two phases of matter, typically a metallic phase (the electrode) and a conductive solution, or electrolyte. Leaving aside the possibility that visiting space folk may have left flashlight batteries or the discoveries of babylonian cells (see panel on electrochemical archaeology), electrochemistry is 200 years old (1791 1991). Electrochemistry is the study of chemical processes that cause electrons to move. this movement of electrons is called electricity, which can be generated by movements of electrons from one element to another in a reaction known as an oxidation reduction ("redox") reaction. The world of biology involves many redox reactions, as well as ion transport through membranes and electrical conduction in the nervous system. this appendix is intended to serve as an introduction to the basic concepts underlying electrochemistry and their linkages to thermodynamics.
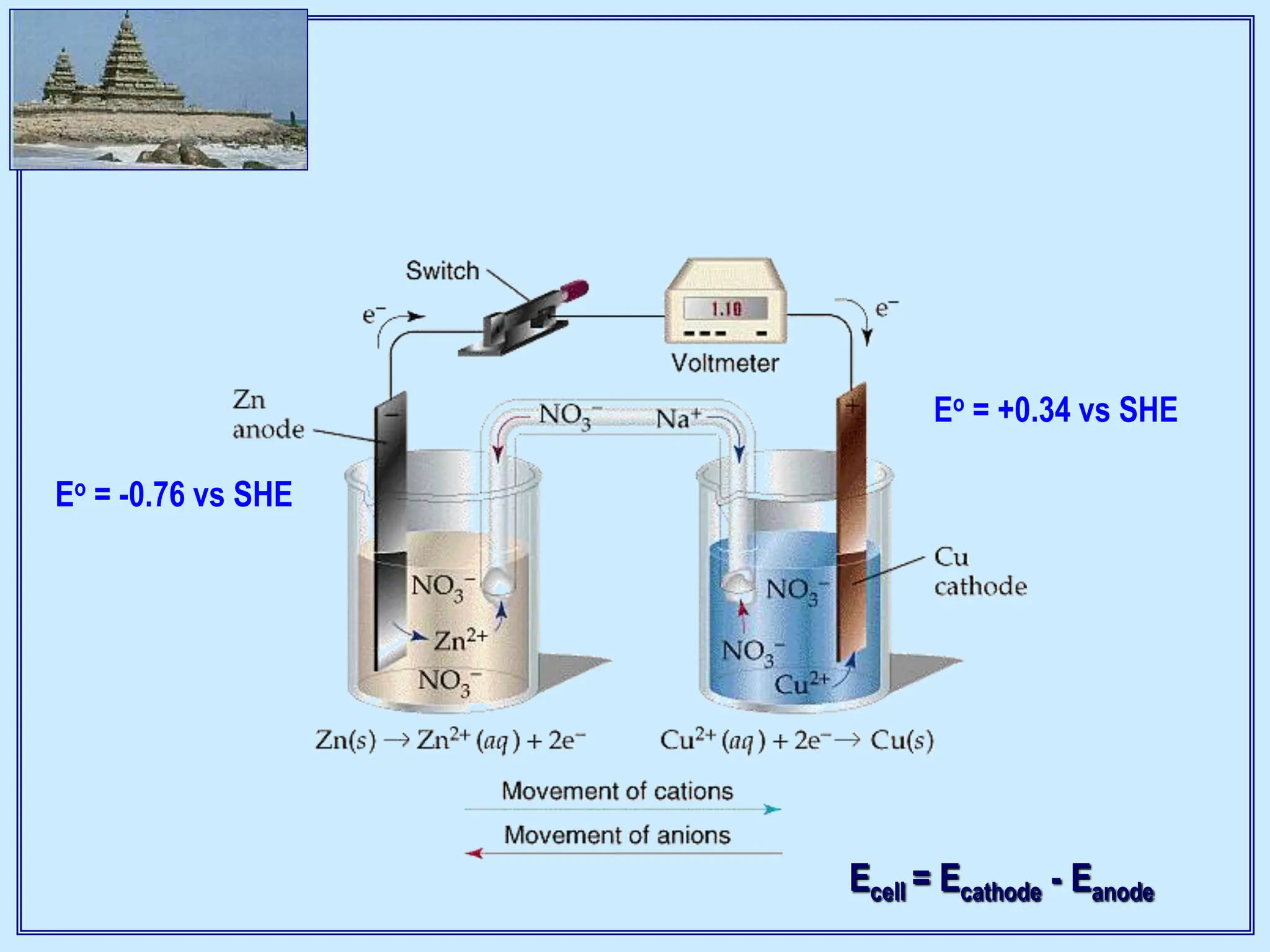
I Electrochemistry Introduction 2014 15 Class 1 And 2 Ppt Electrochemistry is the study of chemical processes that cause electrons to move. this movement of electrons is called electricity, which can be generated by movements of electrons from one element to another in a reaction known as an oxidation reduction ("redox") reaction. The world of biology involves many redox reactions, as well as ion transport through membranes and electrical conduction in the nervous system. this appendix is intended to serve as an introduction to the basic concepts underlying electrochemistry and their linkages to thermodynamics.

Chapter 18 Electrochemistry Pdf Redox Electrochemistry
Comments are closed.