Intro To Catalysis

Catalysis Network Blog Catalysis is the ability of some species to rapidly speed up the rate at which a chemical reaction proceeds. for historical reasons, the discipline is normally split into two sub catagories; homogeneous (homo = same, geneous = phase) and heterogeneous (hetero = different). There are three subdisciplines in catalysis: heterogeneous catalysis (solid state catalysts), homogeneous catalysis, and biocatalysis. in the following sections we will explain two additional concepts of single atom catalysis (sac) and green catalytic processes.

Chapter 1 Intro Pdf Pdf Catalysis Chemical Reactor Catalysis in chemistry is defined as the process in which the rate of the reaction is influenced by the presence of some specific substance. these specific substances are called catalysts. the catalyst is never consumed during the chemical reaction. Catalysis, the modification of the rate of a chemical reaction, usually an acceleration, by addition of a substance not consumed during the reaction. each catalyst molecule may induce the transformation of many molecules of reactants. Among the factors affecting chemical reaction rates discussed earlier in this chapter was the presence of a catalyst, a substance that can increase the reaction rate without being consumed in the reaction. Catalysts affect the rate of a chemical reaction by altering its mechanism to provide a lower activation energy. catalysts can be homogenous (in the same phase as the reactants) or heterogeneous (a different phase than the reactants).

Homogeneouscatalysis Intro Pdf Hydrogenation Catalysis Among the factors affecting chemical reaction rates discussed earlier in this chapter was the presence of a catalyst, a substance that can increase the reaction rate without being consumed in the reaction. Catalysts affect the rate of a chemical reaction by altering its mechanism to provide a lower activation energy. catalysts can be homogenous (in the same phase as the reactants) or heterogeneous (a different phase than the reactants). Catalysis is the process by which a substance, called a catalyst, increases the rate of a chemical reaction without being consumed or altered itself . Introduction to catalysis a catalyst is a compound that allows a reaction that does not proceed in its absence. it is added to the reaction mixture in quantities that are much lower than stoichio metric ones (between 10–6 and 10–1) and, in principle, it is found unchanged at the end of the reaction. A catalyst participates in a reaction but it is neither consumed nor produced (it is not part of the net reaction). a catalyst must be returned to its original condition by the end of the catalytic cycle, so that it can continue to participate in the reaction. Ap chem guide's crash course on catalysis.

Catalysis Intro 1 Pptx Catalysis is the process by which a substance, called a catalyst, increases the rate of a chemical reaction without being consumed or altered itself . Introduction to catalysis a catalyst is a compound that allows a reaction that does not proceed in its absence. it is added to the reaction mixture in quantities that are much lower than stoichio metric ones (between 10–6 and 10–1) and, in principle, it is found unchanged at the end of the reaction. A catalyst participates in a reaction but it is neither consumed nor produced (it is not part of the net reaction). a catalyst must be returned to its original condition by the end of the catalytic cycle, so that it can continue to participate in the reaction. Ap chem guide's crash course on catalysis.
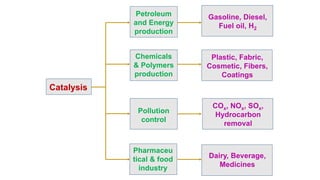
Catalysis Intro 1 Pptx A catalyst participates in a reaction but it is neither consumed nor produced (it is not part of the net reaction). a catalyst must be returned to its original condition by the end of the catalytic cycle, so that it can continue to participate in the reaction. Ap chem guide's crash course on catalysis.

Catalysis Intro 1 Pptx
Comments are closed.