Intro To Bohrs Model Of The Hydrogen Like Atom 3 Of 3 Doc Physics

Bohr Model Of Hydrogen Atom Unifyphysics Bohr’s model of the hydrogen atom, proposed by niels bohr in 1913, was the first quantum model that correctly explained the hydrogen emission spectrum. bohr’s model combines the classical mechanics of planetary motion with the quantum concept of photons. Bohr’s model of the hydrogen atom, proposed by niels bohr in 1913, was the first quantum model that correctly explained the hydrogen emission spectrum. bohr’s model combines the classical mechanics of planetary motion with the quantum concept of photons.

Solution Bohrs Model For Hydrogen Atom Studypool Finally, bohr's model makes a prediction that can be tested! and it's good! it just makes no sense at all. Bohr’s theory explained the atomic spectrum of hydrogen and established new and broadly applicable principles in quantum mechanics. figure 1. niels bohr, danish physicist, used the planetary model of the atom to explain the atomic spectrum and size of the hydrogen atom. Bohr's theory of the hydrogen atom revolutionized our understanding of atomic structure. it explained the discrete nature of atomic spectra and introduced the concept of quantized energy levels, paving the way for modern quantum mechanics. Bohr’s theory explained the atomic spectrum of hydrogen and established new and broadly applicable principles in quantum mechanics. figure 1. niels bohr, danish physicist, used the planetary model of the atom to explain the atomic spectrum and size of the hydrogen atom.

Solution Bohrs Model For Hydrogen Atom Studypool Bohr's theory of the hydrogen atom revolutionized our understanding of atomic structure. it explained the discrete nature of atomic spectra and introduced the concept of quantized energy levels, paving the way for modern quantum mechanics. Bohr’s theory explained the atomic spectrum of hydrogen and established new and broadly applicable principles in quantum mechanics. figure 1. niels bohr, danish physicist, used the planetary model of the atom to explain the atomic spectrum and size of the hydrogen atom. Ncert class 11 chemistry 2.4: bohr’s model for hydrogen atom – quantized orbits, angular momentum, energy levels, and how bohr explained hydrogen’s line spectrum with limitations. Bohr’s theory explained the atomic spectrum of hydrogen and established new and broadly applicable principles in quantum mechanics. figure 1. niels bohr, danish physicist, used the planetary model of the atom to explain the atomic spectrum and size of the hydrogen atom. Bohr's model is an atomic model proposed by danish physicist niels bohr in 1913. in an atom, the electrons revolve around the nucleus in definite energy levels called orbits shells. this model provides a basic understanding of the concept of the atom and its constituents. Bohr’s theory explained the atomic spectrum of hydrogen and established new and broadly applicable principles in quantum mechanics. niels bohr, danish physicist, used the planetary model of the atom to explain the atomic spectrum and size of the hydrogen atom.
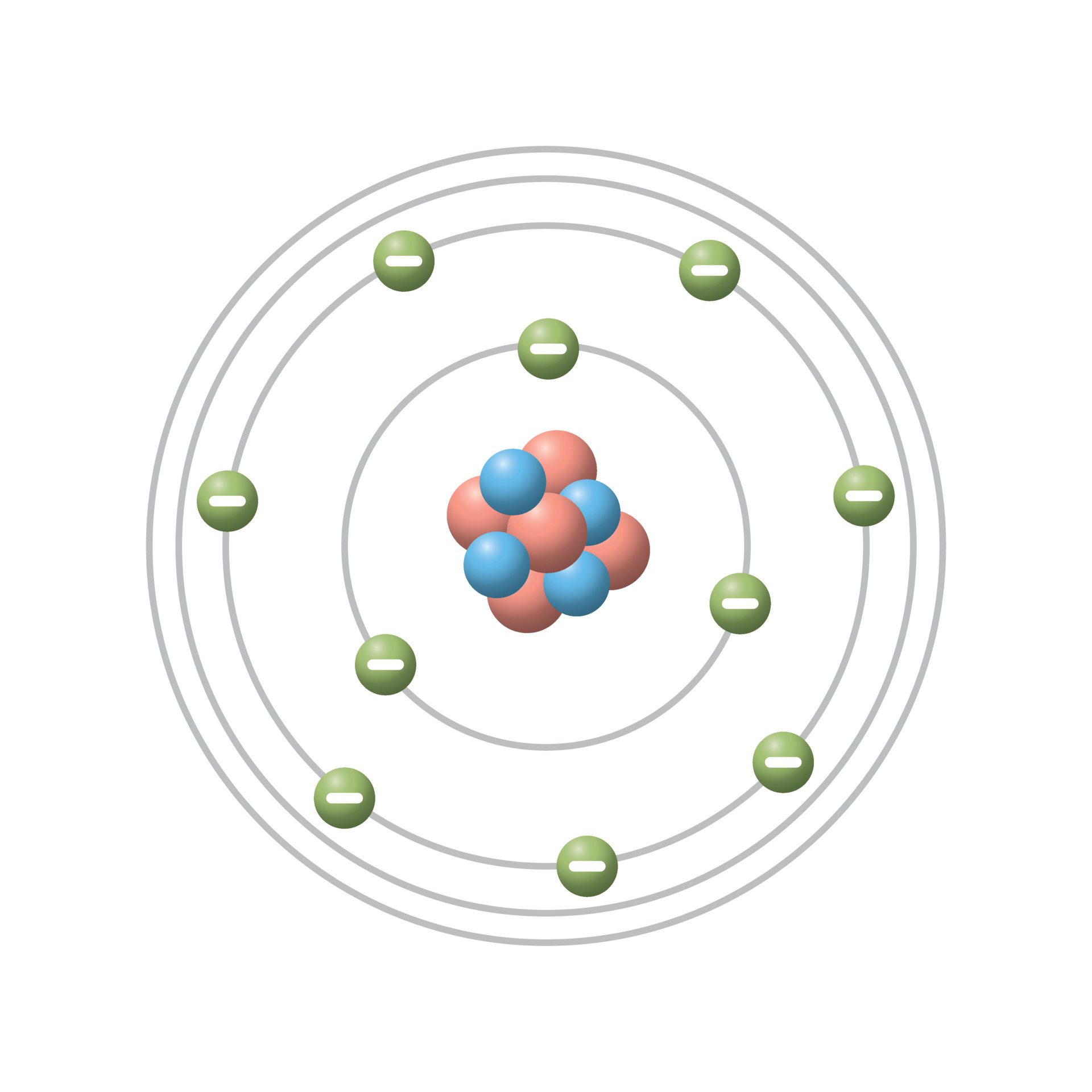
Bohr S Model Of Atom Tolfarea Ncert class 11 chemistry 2.4: bohr’s model for hydrogen atom – quantized orbits, angular momentum, energy levels, and how bohr explained hydrogen’s line spectrum with limitations. Bohr’s theory explained the atomic spectrum of hydrogen and established new and broadly applicable principles in quantum mechanics. figure 1. niels bohr, danish physicist, used the planetary model of the atom to explain the atomic spectrum and size of the hydrogen atom. Bohr's model is an atomic model proposed by danish physicist niels bohr in 1913. in an atom, the electrons revolve around the nucleus in definite energy levels called orbits shells. this model provides a basic understanding of the concept of the atom and its constituents. Bohr’s theory explained the atomic spectrum of hydrogen and established new and broadly applicable principles in quantum mechanics. niels bohr, danish physicist, used the planetary model of the atom to explain the atomic spectrum and size of the hydrogen atom.
Comments are closed.