Informed Consent For Medical Research Case Studies Pdf Informed

Informed Consent For Medical Research Case Studies Pdf Informed This document summarizes a research article about informed consent for medical research. it discusses two case studies involving challenges with obtaining informed consent from patients in an emergency department setting. Issues with informed consent in the medical care arena preceded the larger scale devastating breaches of informed con sent in medical research, and began to form the groundwork for many of the laws governing medical data today.

Informed Consent In Research Types Templates And Examples This paper provides a comprehensive review of the tenets underlying informed consent in research, including the assessment of capacity to consent, considerations for patients unable to consent, when to seek consent from substitute decision makers, and consent under special circumstances. To assist researchers, who has developed informed consent form templates for various types of research studies. click here to view these. Pdf | on may 19, 2025, matea valešić and others published informed consent and ethics committee involvement in case reports and case series: cross sectional meta research study | find,. Before sharing human biospecimens or private data, it is essential to check with the irb approved informed consent document to determine whether and exactly what sharing is permitted.

Informed Research Consent Form Fillable Pdf Template Pdf | on may 19, 2025, matea valešić and others published informed consent and ethics committee involvement in case reports and case series: cross sectional meta research study | find,. Before sharing human biospecimens or private data, it is essential to check with the irb approved informed consent document to determine whether and exactly what sharing is permitted. The article examines the three essential features of informed consent, patient capacity, disclosure of information, and voluntariness, and a review of five exceptions to the duty to obtain informed consent. The consent discussion and the consent form must contain information to allow prospective subjects to make an informed decision about participation in a clinical investigation and provide. Modern research ethics frameworks, such as the declaration of helsinki and the belmont report, emphasize the central role of informed consent in protecting participants. The fact sheets deal with aspects directly related to the informed consent process, while the tools include aspects that do not strictly belong to the informed consent process but are useful for its better development.
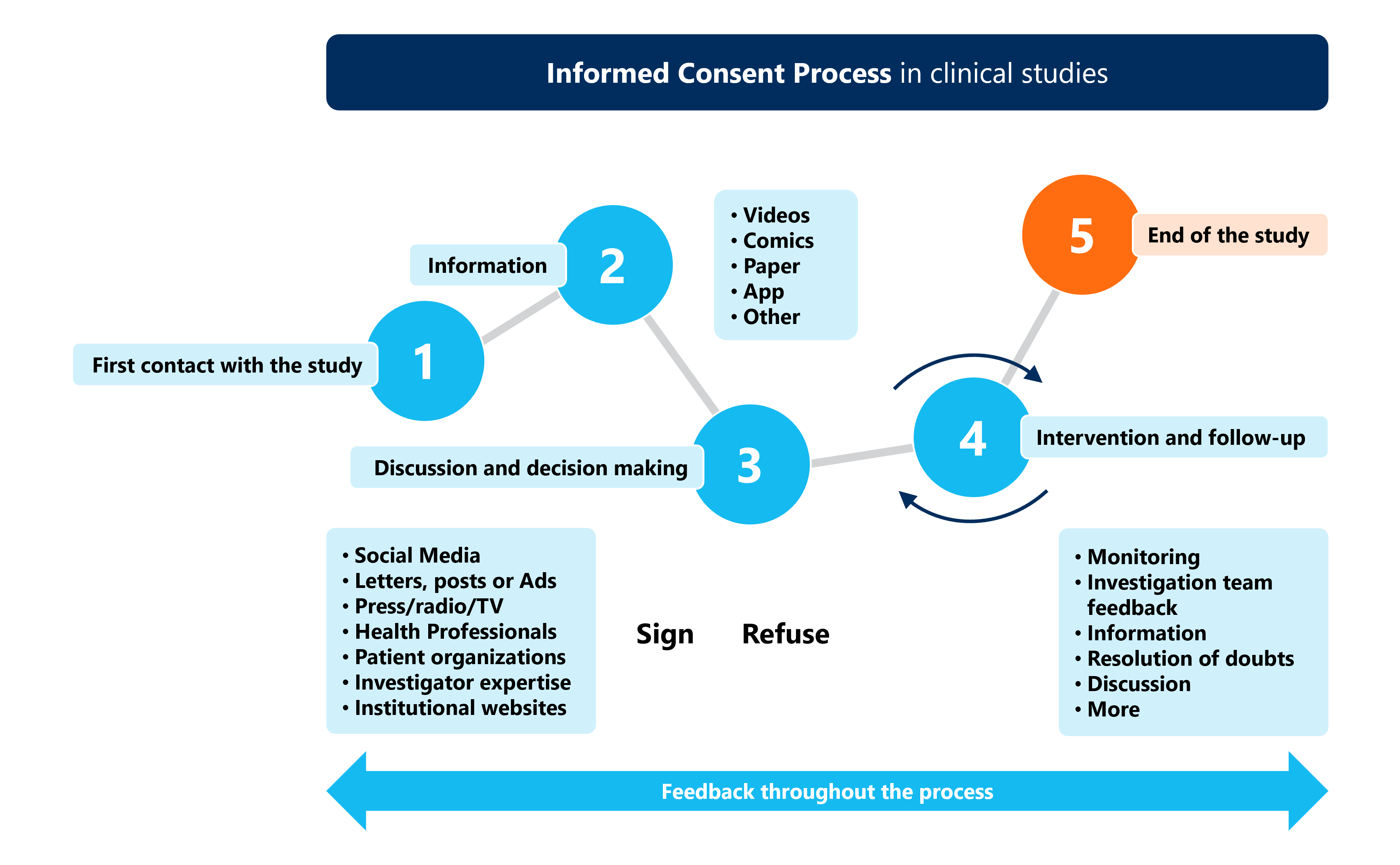
Informed Consent In Clinical Research Ethical Essentials The article examines the three essential features of informed consent, patient capacity, disclosure of information, and voluntariness, and a review of five exceptions to the duty to obtain informed consent. The consent discussion and the consent form must contain information to allow prospective subjects to make an informed decision about participation in a clinical investigation and provide. Modern research ethics frameworks, such as the declaration of helsinki and the belmont report, emphasize the central role of informed consent in protecting participants. The fact sheets deal with aspects directly related to the informed consent process, while the tools include aspects that do not strictly belong to the informed consent process but are useful for its better development.
Comments are closed.