Immune Checkpoint Inhibitor Toxicities

Fatal Toxicities Vary By Immune Checkpoint Inhibitor Type Though effective, ici use is accompanied by a milieu of potentially bothersome or even life threatening toxicities known as immune related adverse events (iraes), necessitating careful monitoring and early intervention. Earn cme ce and moc credits for using the nccn guidelines to research your patient care decisions and clinical questions by clicking here.
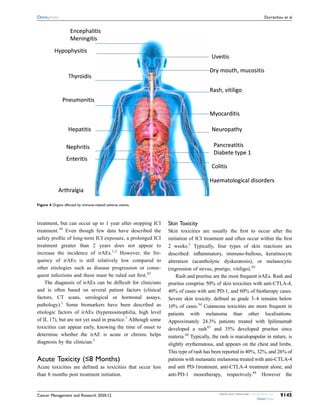
Management Of Immune Checkpoint Inhibitor Toxicities Pdf Immune checkpoint inhibitors (icis), also known as checkpoint inhibitor immunotherapy, are immunomodulatory antibodies that are used to enhance the immune system. Immune checkpoint inhibitors (icis) produce often durable responses in many cancer types but are associated with autoimmune like toxicities. these immune related adverse events (iraes) occur in multiple organ systems and often improve with steroids or therapy cessation. In this review, we describe current preclinical models and immune implications of ici induced skin toxicities, colitis, neurological and endocrine toxicities, pneumonitis, arthritis, and myocarditis along with their management. Immune checkpoint inhibitors (ici) have revolutionized cancer treatment by harnessing the immune system but ici can induce life threatening immune related adverse events (irae) affecting every organ.

Nccn 2025 Managing Immune Checkpoint Inhibitor Related Toxicities In In this review, we describe current preclinical models and immune implications of ici induced skin toxicities, colitis, neurological and endocrine toxicities, pneumonitis, arthritis, and myocarditis along with their management. Immune checkpoint inhibitors (ici) have revolutionized cancer treatment by harnessing the immune system but ici can induce life threatening immune related adverse events (irae) affecting every organ. Eochagain, c. m. et al. management of immune checkpoint inhibitor associated toxicities in older adults with cancer: recommendations from the international society of geriatric oncology (siog). Icis act by modulating the immune system to attack cancer cells through inhibition of cytotoxic t lymphocyte–associated antigen 4 (ctla 4), programmed death 1 (pd 1), or programmed death ligand 1 (pd l1), so their potential toxicities are distinct from those of cytotoxic chemotherapy. This clinical practice guideline focuses on one overarching clinical question: how should clinicians manage immune mediated adverse events (aes) in adult patients with cancer treated with immune checkpoint blockade antibodies?. In this review, we discuss our current understanding of the inflammatory toxicities from immune checkpoint inhibitors (icis) and propose optimal treatment strategies for these toxicities.

Pdf Management Of Immune Checkpoint Inhibitor Toxicities A Review Eochagain, c. m. et al. management of immune checkpoint inhibitor associated toxicities in older adults with cancer: recommendations from the international society of geriatric oncology (siog). Icis act by modulating the immune system to attack cancer cells through inhibition of cytotoxic t lymphocyte–associated antigen 4 (ctla 4), programmed death 1 (pd 1), or programmed death ligand 1 (pd l1), so their potential toxicities are distinct from those of cytotoxic chemotherapy. This clinical practice guideline focuses on one overarching clinical question: how should clinicians manage immune mediated adverse events (aes) in adult patients with cancer treated with immune checkpoint blockade antibodies?. In this review, we discuss our current understanding of the inflammatory toxicities from immune checkpoint inhibitors (icis) and propose optimal treatment strategies for these toxicities.

Immune Related Adverse Events Of Immune Checkpoint Inhibitors For This clinical practice guideline focuses on one overarching clinical question: how should clinicians manage immune mediated adverse events (aes) in adult patients with cancer treated with immune checkpoint blockade antibodies?. In this review, we discuss our current understanding of the inflammatory toxicities from immune checkpoint inhibitors (icis) and propose optimal treatment strategies for these toxicities.
Comments are closed.