Hydroxyl Group Part 2 Examples

Chapter 1 Hydroxyl Group Chm132 Pdf Today's video is just a recap of the last video (linked below). i made a few clarifications, and also provided a few examples of groups that contain hydroxyl groups. hope you enjoy!. What is a hydroxyl group. learn its formula, structure, properties, preparations, reactions, and uses. check out a few examples of compounds containing hydroxyl.
Hydroxyl Group Examples Like other compounds, those with multiple hydroxyl groups are named in a similar way; the prefix indicates the number of hydroxyl groups. this is illustrated by the fact that, for instance, a compound which contains two hydroxyl groups will be a "diol" and one which has three will be a "triol.". Hydroxyl group (―oh), in chemistry, a functional group with one hydrogen and one oxygen atom. an oxygen atom normally forms two σ bonds with other atoms; the water molecule, h2o, is the simplest and most common example. A hydroxy or hydroxyl group is a functional group composed of one oxygen atom covalently bonded to one hydrogen atom and having the chemical formula oh. compounds such as alcohols, phenols, and carboxylic acids contain these groups. Hydroxyl group examples: examples of compounds containing hydroxyl groups include ethanol (alcoholic beverages), sucrose (table sugar), glycerol (in fats and oils), and various other compounds like phenol, propanol, methanol, and ethylene glycol.
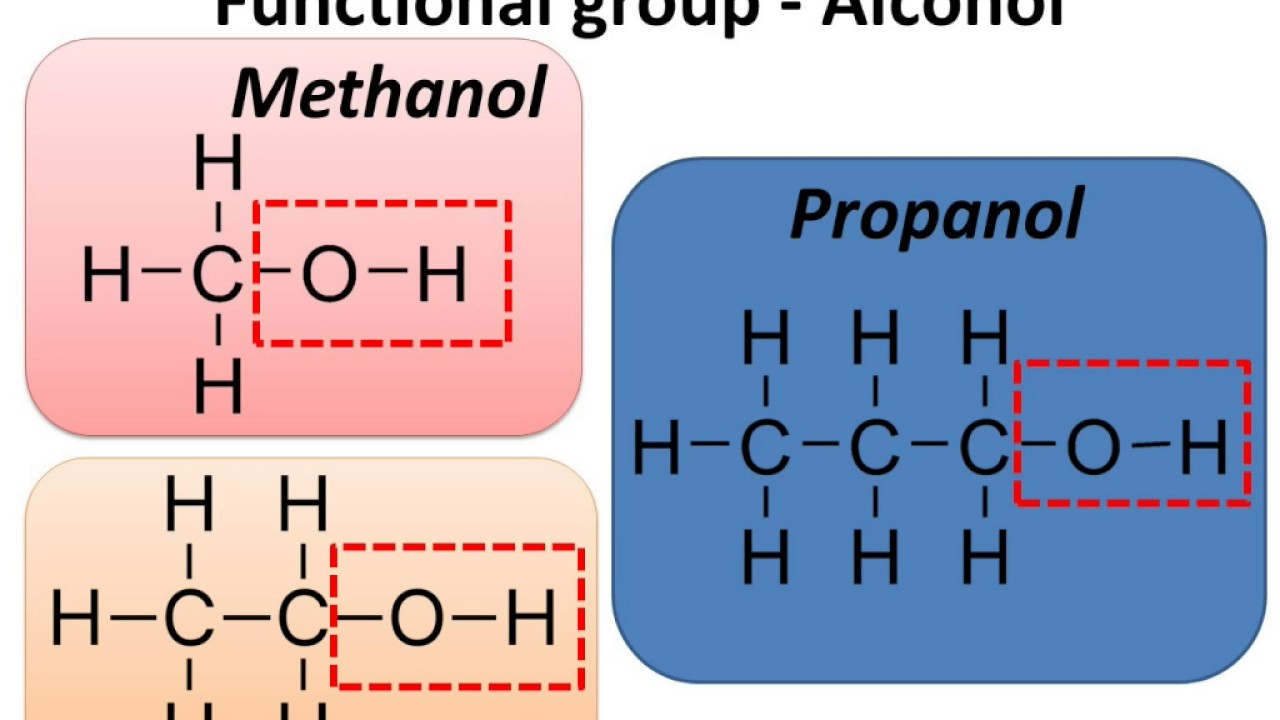
Hydroxyl Group Examples Anomers Biochemistry Flashcards Draw It To A hydroxy or hydroxyl group is a functional group composed of one oxygen atom covalently bonded to one hydrogen atom and having the chemical formula oh. compounds such as alcohols, phenols, and carboxylic acids contain these groups. Hydroxyl group examples: examples of compounds containing hydroxyl groups include ethanol (alcoholic beverages), sucrose (table sugar), glycerol (in fats and oils), and various other compounds like phenol, propanol, methanol, and ethylene glycol. Hydroxyl group is a functional group which has the chemical formula oh and consists of an oxygen atom covalently bonded to a hydrogen atom. it is found in various organic compounds. both alcohols and carboxylic acids contain one or more hydroxyl groups, which are essential in organic chemistry. Learn the molecule of hydroxyl and the properties of hydroxyl molecules that can act as a functional group. see the structure and examples in compounds. Hydroxyl group (substance class: alcohol, suffix ol) in a hydroxyl group ( oh), a hydrogen atom is bonded to an oxygen atom. this in turn is linked to a carbon atom of the backbone of an organic molecule. note ( oh s oh ). example: ethanol (=drinking alcohol). The presence of a highly electronegative group allows alcohols to hydrogen bond with other molecules. for example, in ethanol, the negatively charged oxygen on one ethanol molecule is attracted to a positively charged hydrogen molecule on another ethanol molecule.

Hydroxyl Group Examples Anomers Biochemistry Flashcards Draw It To Hydroxyl group is a functional group which has the chemical formula oh and consists of an oxygen atom covalently bonded to a hydrogen atom. it is found in various organic compounds. both alcohols and carboxylic acids contain one or more hydroxyl groups, which are essential in organic chemistry. Learn the molecule of hydroxyl and the properties of hydroxyl molecules that can act as a functional group. see the structure and examples in compounds. Hydroxyl group (substance class: alcohol, suffix ol) in a hydroxyl group ( oh), a hydrogen atom is bonded to an oxygen atom. this in turn is linked to a carbon atom of the backbone of an organic molecule. note ( oh s oh ). example: ethanol (=drinking alcohol). The presence of a highly electronegative group allows alcohols to hydrogen bond with other molecules. for example, in ethanol, the negatively charged oxygen on one ethanol molecule is attracted to a positively charged hydrogen molecule on another ethanol molecule.
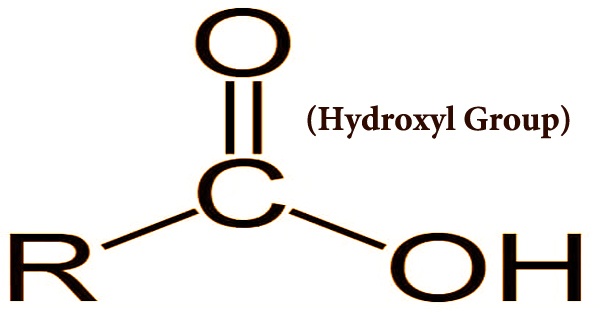
Hydroxyl Group Assignment Point Hydroxyl group (substance class: alcohol, suffix ol) in a hydroxyl group ( oh), a hydrogen atom is bonded to an oxygen atom. this in turn is linked to a carbon atom of the backbone of an organic molecule. note ( oh s oh ). example: ethanol (=drinking alcohol). The presence of a highly electronegative group allows alcohols to hydrogen bond with other molecules. for example, in ethanol, the negatively charged oxygen on one ethanol molecule is attracted to a positively charged hydrogen molecule on another ethanol molecule.

Hydroxyl Group Webref Org
Comments are closed.