Hydroxyl Group Examples
Hydroxyl Group Examples What is a hydroxyl group. learn its formula, structure, properties, preparations, reactions, and uses. check out a few examples of compounds containing hydroxyl. Like other compounds, those with multiple hydroxyl groups are named in a similar way; the prefix indicates the number of hydroxyl groups. this is illustrated by the fact that, for instance, a compound which contains two hydroxyl groups will be a "diol" and one which has three will be a "triol.".
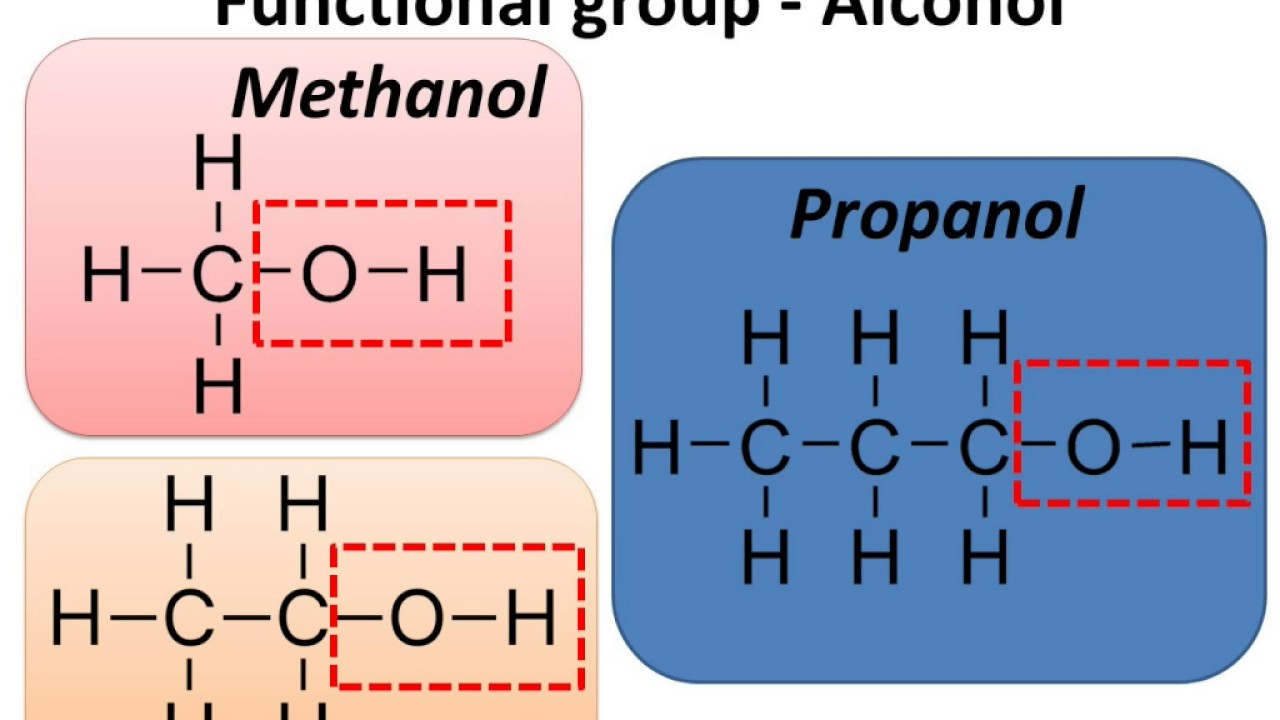
Hydroxyl Group Examples Anomers Biochemistry Flashcards Draw It To The hydroxyl group is referred to as a hydrophilic (“water loving”) group, because it forms hydrogen bonds with water and enhances the solubility of an alcohol in water. methanol, ethanol, n propyl alcohol, isopropyl alcohol, and t butyl alcohol are all miscible with water. In chemistry, a hydroxy or hydroxyl group is a functional group with the chemical formula −oh and composed of one oxygen atom covalently bonded to one hydrogen atom. in organic chemistry, alcohols and carboxylic acids contain one or more hydroxy groups. A hydroxy or hydroxyl group is a functional group composed of one oxygen atom covalently bonded to one hydrogen atom and having the chemical formula oh. compounds such as alcohols, phenols, and carboxylic acids contain these groups. Hydroxyl group examples: examples of compounds containing hydroxyl groups include ethanol (alcoholic beverages), sucrose (table sugar), glycerol (in fats and oils), and various other compounds like phenol, propanol, methanol, and ethylene glycol.
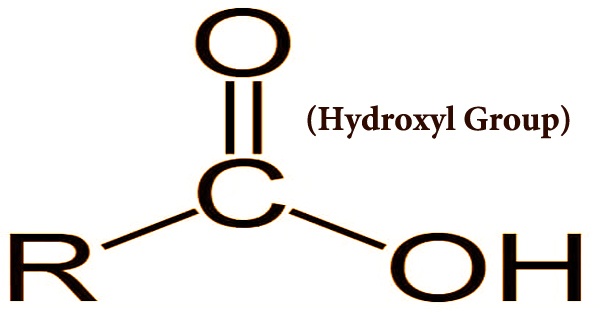
Hydroxyl Group Assignment Point A hydroxy or hydroxyl group is a functional group composed of one oxygen atom covalently bonded to one hydrogen atom and having the chemical formula oh. compounds such as alcohols, phenols, and carboxylic acids contain these groups. Hydroxyl group examples: examples of compounds containing hydroxyl groups include ethanol (alcoholic beverages), sucrose (table sugar), glycerol (in fats and oils), and various other compounds like phenol, propanol, methanol, and ethylene glycol. Water, with its simple h oh structure, is the most abundant and well known compound featuring a hydroxyl group. carbohydrates, including various sugars like glucose, are also defined by the presence of multiple hydroxyl groups, often referred to as polyols, which contribute to their solubility. Alcohol is an example of a molecule that contains a hydroxyl group. ethanol, methanol, isopropanol are some other examples of alcohol. a hydroxyl group is composed of one atom of hydrogen and one. Monosaccharides and disaccharides contain multiple hydroxyl groups that contribute to their reactivity, especially in glycosidic bond formation during carbohydrate reactions. Hydroxyl group is a functional group which has the chemical formula oh and consists of an oxygen atom covalently bonded to a hydrogen atom. it is found in various organic compounds. both alcohols and carboxylic acids contain one or more hydroxyl groups, which are essential in organic chemistry.

Hydroxyl Group Webref Org Water, with its simple h oh structure, is the most abundant and well known compound featuring a hydroxyl group. carbohydrates, including various sugars like glucose, are also defined by the presence of multiple hydroxyl groups, often referred to as polyols, which contribute to their solubility. Alcohol is an example of a molecule that contains a hydroxyl group. ethanol, methanol, isopropanol are some other examples of alcohol. a hydroxyl group is composed of one atom of hydrogen and one. Monosaccharides and disaccharides contain multiple hydroxyl groups that contribute to their reactivity, especially in glycosidic bond formation during carbohydrate reactions. Hydroxyl group is a functional group which has the chemical formula oh and consists of an oxygen atom covalently bonded to a hydrogen atom. it is found in various organic compounds. both alcohols and carboxylic acids contain one or more hydroxyl groups, which are essential in organic chemistry.

Hydroxyl Group Monosaccharides and disaccharides contain multiple hydroxyl groups that contribute to their reactivity, especially in glycosidic bond formation during carbohydrate reactions. Hydroxyl group is a functional group which has the chemical formula oh and consists of an oxygen atom covalently bonded to a hydrogen atom. it is found in various organic compounds. both alcohols and carboxylic acids contain one or more hydroxyl groups, which are essential in organic chemistry.
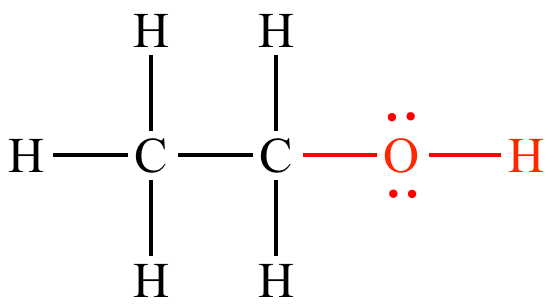
Illustrated Glossary Of Organic Chemistry Hydroxyl Group
Comments are closed.