Hydroxyl Functional Group Explained

Hydroxyl Functional Group Hydroxyl group (―oh), in chemistry, a functional group with one hydrogen and one oxygen atom. an oxygen atom normally forms two σ bonds with other atoms; the water molecule, h2o, is the simplest and most common example. Hydroxyl is an chemical group that plays a big role in organic chemistry. it is an oh functional group with an oxygen atom bonded to a hydrogen atom, and it is usually represented as oh.
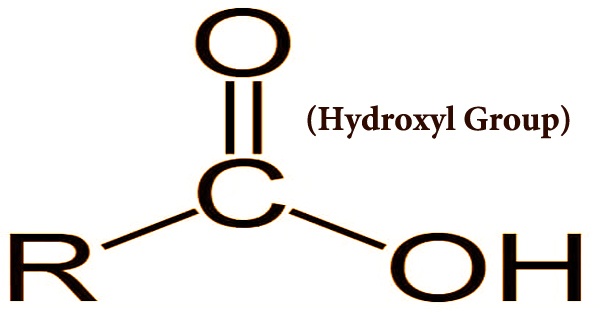
Hydroxyl Group Assignment Point What is a hydroxyl group. learn its formula, structure, properties, preparations, reactions, and uses. check out a few examples of compounds containing hydroxyl. In chemistry, a hydroxy or hydroxyl group is a functional group with the chemical formula −oh and composed of one oxygen atom covalently bonded to one hydrogen atom. in organic chemistry, alcohols and carboxylic acids contain one or more hydroxy groups. Hydroxyl groups are functional groups commonly found in organic molecules, including alcohols, sugars, and carboxylic acids. they are composed of an oxygen atom and a hydrogen atom, which are covalently bonded to a carbon atom. The hydroxyl group, represented as oh, is a stable arrangement of one oxygen atom covalently bonded to one hydrogen atom. this functional group is commonly found within larger organic molecules, where its presence profoundly influences molecular characteristics.

The Hydroxyl Functional Group Pdf Functional Group Chemical Polarity Hydroxyl groups are functional groups commonly found in organic molecules, including alcohols, sugars, and carboxylic acids. they are composed of an oxygen atom and a hydrogen atom, which are covalently bonded to a carbon atom. The hydroxyl group, represented as oh, is a stable arrangement of one oxygen atom covalently bonded to one hydrogen atom. this functional group is commonly found within larger organic molecules, where its presence profoundly influences molecular characteristics. Learn the molecule of hydroxyl and the properties of hydroxyl molecules that can act as a functional group. see the structure and examples in compounds. The hydroxyl group is a functional group consisting of a hydrogen atom covalently bonded to an oxygen atom. the hydroxyl group is denoted by oh in chemical structures and has a valence charge of 1. A hydroxyl group is a functional group characterized by the presence of an oxygen atom bonded to a hydrogen atom, represented as oh. this polar group plays. A hydroxyl group is a functional group consisting of one oxygen atom and one hydrogen atom, chemically denoted as oh. it forms part of various organic compounds, notably alcohols.

Functional Groups Hydroxyl The Explanation Says That A Higher Ph Will Learn the molecule of hydroxyl and the properties of hydroxyl molecules that can act as a functional group. see the structure and examples in compounds. The hydroxyl group is a functional group consisting of a hydrogen atom covalently bonded to an oxygen atom. the hydroxyl group is denoted by oh in chemical structures and has a valence charge of 1. A hydroxyl group is a functional group characterized by the presence of an oxygen atom bonded to a hydrogen atom, represented as oh. this polar group plays. A hydroxyl group is a functional group consisting of one oxygen atom and one hydrogen atom, chemically denoted as oh. it forms part of various organic compounds, notably alcohols.
Comments are closed.