Hydrogen Atom Bohr Model

Hydrogen Atom Bohr Model Cartoon Vector Cartoondealer 267662053 In 1921, following the work of chemists and others involved in work on the periodic table, bohr extended the model of hydrogen to give an approximate model for heavier atoms. Bohr’s model of the hydrogen atom, proposed by niels bohr in 1913, was the first quantum model that correctly explained the hydrogen emission spectrum. bohr’s model combines the classical mechanics of planetary motion with the quantum concept of photons.
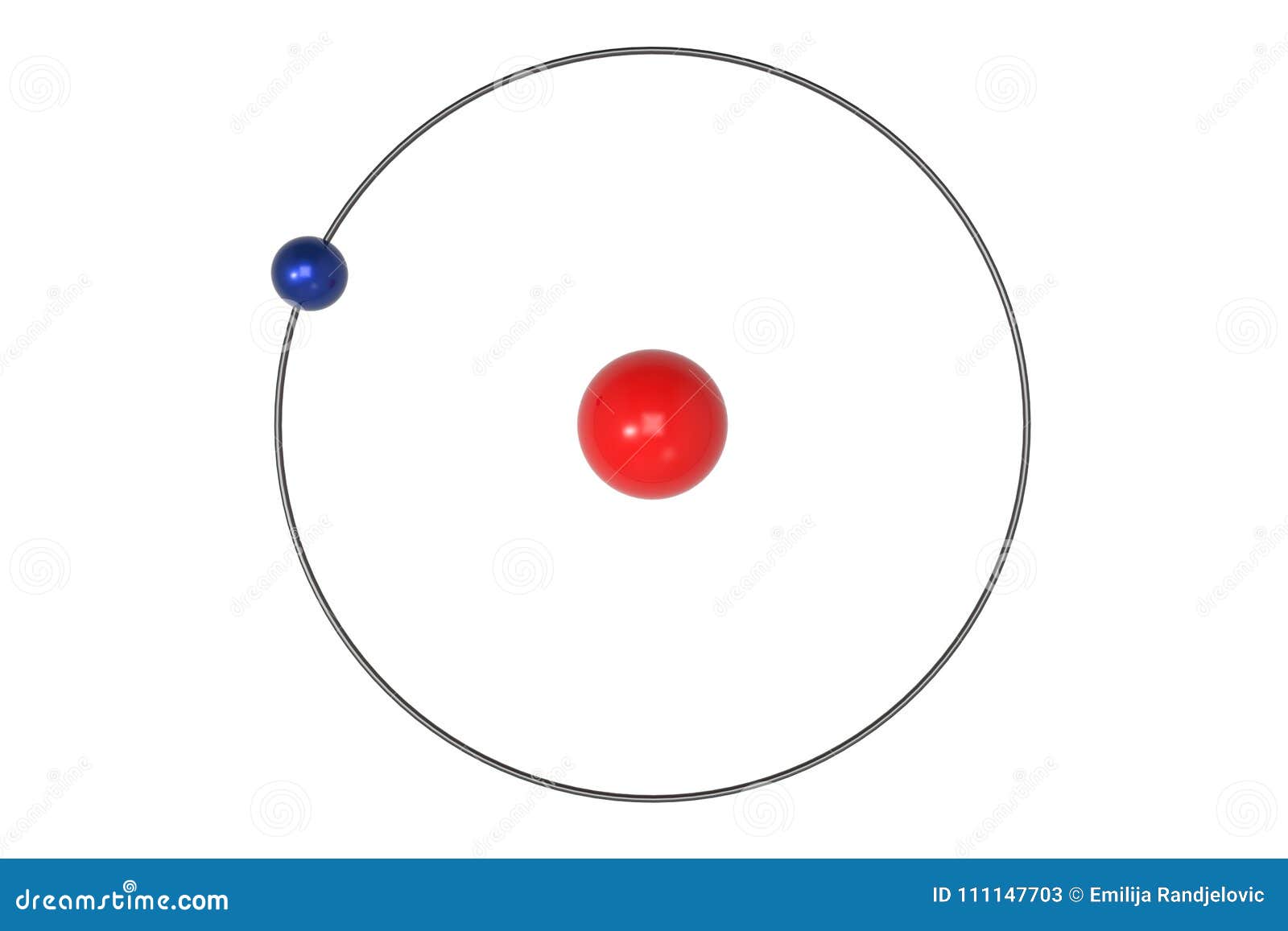
Bohr Model Hydrogen Atom My Xxx Hot Girl Try out different models by shooting light at the atom. check how the prediction of the model matches the experimental results. What does the bohr model explain? the bohr model could account for the series of discrete wavelengths in the emission spectrum of hydrogen. niels bohr proposed that light radiated from hydrogen atoms only when an electron made a transition from an outer orbit to one closer to the nucleus. The bohr model, often referred to as the planetary model of the atom, is a concept that simplifies the complex nature of atomic structures. it’s particularly focused on the hydrogen atom, which is the simplest atom with only one electron. Bohr’s model was a very successful and important contribution to the studies of atomic structure and explaining the line spectrum of hydrogen. in fact, bohr was awarded the nobel prize in physics in 1922.
Bohr Model Of Hydrogen Atom Royalty Free Stock Photography The bohr model, often referred to as the planetary model of the atom, is a concept that simplifies the complex nature of atomic structures. it’s particularly focused on the hydrogen atom, which is the simplest atom with only one electron. Bohr’s model was a very successful and important contribution to the studies of atomic structure and explaining the line spectrum of hydrogen. in fact, bohr was awarded the nobel prize in physics in 1922. Bohr was the first to recognize this by incorporating the idea of quantization into the electronic structure of the hydrogen atom, and he was able to thereby explain the emission spectra of hydrogen as well as other one electron systems. Bohr’s theory explained the atomic spectrum of hydrogen and established new and broadly applicable principles in quantum mechanics. figure 1. niels bohr, danish physicist, used the planetary model of the atom to explain the atomic spectrum and size of the hydrogen atom. Bohr's model of the hydrogen atom was the first to incorporate quantum theory, and the key idea of his model was that electrons occupy discrete orbitals. the bohr model of the atom was proposed by niels bohr in 1913 as an expansion on and correction of the rutherford model. The bohr model of the hydrogen atom explains the connection between the quantization of photons and the quantized emission from atoms. bohr described the hydrogen atom in terms of an electron moving in a circular orbit about a nucleus.
Comments are closed.